To draft recommendations on interleukin 6 (IL-6) blockade in rheumatoid arthritis (RA), based on best evidence and experience.
MethodsA group of 10 experts on IL-6 blockade in RA was selected. The 2 coordinators formulated 23 questions about IL-6 blockade (indications, efficacy, safety, etc.). A systematic review was conducted to answer the questions. Using this information, inclusion and exclusion criteria were established, as were the search strategies (Medline, EMBASE and the Cochrane Library were searched). Two different reviewers selected the articles. Evidence tables were created. At the same time, European League Against Rheumatism and American College of Rheumatology abstracts were evaluated. Based on this evidence, the coordinators proposed preliminary recommendations that the experts discussed and voted on in a nominal group meeting. The level of evidence and grade of recommendation were established using the Oxford Centre for Evidence Based Medicine and the level of agreement with the Delphi technique (2 rounds). Agreement was established if at least 80% of the experts voted yes (yes/no).
ResultsThe 8 preliminary recommendations were accepted after the Delphi process. They covered aspects such as the use of these therapies in monotherapy, in combination, in patients with refractory disease or intolerant patients, response evaluation, optimisation and risk management.
ConclusionsThe manuscript aims to solve frequently asked questions and aid in decision making strategies when treating RA patients with IL-6 blockade.
Generar recomendaciones sobre el bloqueo de la interleucina 6 (IL-6) en pacientes con artritis reumatoide (AR), basadas en la mejor evidencia y experiencia.
MétodosSe seleccionó a 10 expertos reumatólogos en el manejo de los inhibidores de la IL-6. Los 2 coordinadores generaron 23 preguntas sobre el bloqueo de la IL-6 en la AR (perfiles de indicación, eficacia, seguridad, etc.) para ser contestadas mediante una revisión sistemática de la literatura. Con base en las preguntas se definieron los criterios de inclusión y exclusión, y las estrategias de búsqueda (para interrogar Medline, Embase y la Cochrane Library). Dos revisores seleccionaron los artículos resultantes de la búsqueda. Se generaron tablas de evidencia. Paralelamente, se evaluaron abstracts de congresos de EULAR y ACR. Con toda esta evidencia los coordinadores propusieron 8 recomendaciones preliminares que se evaluaron, discutieron y votaron en una reunión de grupo nominal con el resto de los expertos. Para cada recomendación se estableció el nivel de evidencia y grado de recomendación, y el grado de acuerdo mediante un Delphi. Se definió acuerdo si al menos el 80% de los participantes contestaban sí a la recomendación (sí o no).
ResultadosLas 8 recomendaciones preliminares se aceptaron tras el Delphi. Abarcan aspectos como su uso en monoterapia, en combinación, en pacientes refractarios o intolerantes, la evaluación de su respuesta, la optimización o la gestión del riesgo.
ConclusionesEste documento pretende resolver algunos interrogantes clínicos habituales y facilitar la toma de decisiones con el bloqueo de la IL-6 en el manejo de la AR.
There have been major changes in the treatment of rheumatoid arthritis (RA) in recent years. New drugs and therapy strategies have completely changed the prognosis of these patients.1–4 The biological drugs have been a key element in this field.5 The first experience was with tumour necrosis factor (TNF) inhibitors, but in recent years drugs with different mechanisms of action have appeared, including the interleukin-6 (IL-6) inhibitors.6–18
IL-6 is a pleiotropic cytokine that plays a central role in the pathogenesis of RA by contributing to the activation of T and B lymphocytes, the stimulation of synoviocytes, the maturation of osteoclasts and the production of acute-phase reactants. RA activity has been correlated with the serum elevation of both IL-6 and its soluble receptor (IL-6R), and therefore its blockade (both IL-6 and IL-6R) is considered a good therapeutic target in treatment of the disease.19,20
Tocilizumab (TCZ) was the first IL-6 inhibitor approved and marketed in Spain, for which there is much short and long-term evidence.7,14–18 The EMA has also approved sarilumab (SAR),9–13 another monoclonal antibody against IL-6R, and there are other IL-6 inhibitors such as olokizumab,21,22 clazakizumab,23 and sirukumab,24,25 at different stages of clinical development; however, the latter in the end will not be registered for RA treatment.
The aim of this paper was to generate recommendations based on the best possible evidence and experience with the use of IL-6 inhibitors in patients with RA.
MethodsThe nominal group and Delphi technique were used to prepare the consensus.26 The document was entirely prepared by distribution of tasks and comments to the parties, with the help of a systematic literature review and an expert methodologist.
Selection of expertsFirst, a group of 10 rheumatologists was formed, with recognised experience in the management of RA patients and the use of IL-6 inhibitors. A Medline search was carried out to identify Spanish rheumatologists with publications on the subject of this paper. Submission of papers to the national congress of the Spanish Society of Rheumatology was also considered. All the results were discussed with the coordinators who, in addition to the aforementioned, considered as experts those who regularly attended specific forums, those considered opinion leaders on this subject, who those had been speakers, etc. Finally, the selection sought to achieve the greatest geographical representativeness.
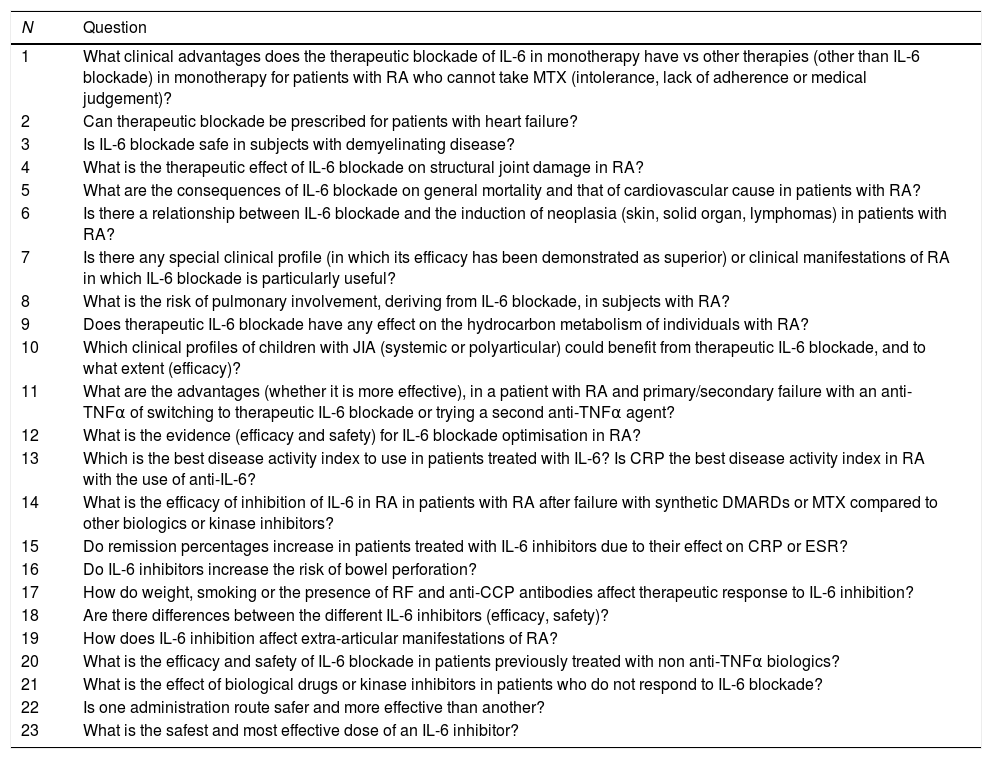
Systematic literature reviewThe project coordinators generated 23 questions to be answered by a systematic review of the literature (Table 1). Since they all referred to different aspects of the same topic, it was decided that one review should be carried out. The inclusion and exclusion criteria were defined with these questions. We searched for articles that included patients with RA under treatment with any IL-6 inhibitor, irrespective of the dose or specific administration route. In addition, these studies had to analyse (depending on the question) different aspects of the efficacy and safety of the drug therapy. In the end, only studies with the following designs were included: meta-analyses, systematic reviews and clinical trials. A search was performed of the literature databases Medline (from its launch until October 2017), Embase (from its launch until October 2017), and the Cochrane Library (from its launch until October 2017). Mesh and free text terms were used, and the search strategy was executed with the help of an expert documentalist. For each review, two reviewers (EL, TO) independently analysed the articles resulting from the search strategy in the different literature databases, and also carried out a detailed analysis of the articles included. In the event of discrepancy, a third person, LC, resolved any conflict. The grey literature in EULAR and ACR congresses over the past 2 years was reviewed as well as documents from the coordinators (articles and abstracts from congresses and other forums of interest, related to the subject of the consensus). Then, a secondary manual search was undertaken of the references of the articles that were eventually included. The Jadad scale was used for the clinical trials (RCT),27 to evaluate the methodological quality of the studies included, Evidence and results tables were created describing the main features and results of the studies included.
Questions formulated to be answered by means of a systematic review of the literature.
| N | Question |
|---|---|
| 1 | What clinical advantages does the therapeutic blockade of IL-6 in monotherapy have vs other therapies (other than IL-6 blockade) in monotherapy for patients with RA who cannot take MTX (intolerance, lack of adherence or medical judgement)? |
| 2 | Can therapeutic blockade be prescribed for patients with heart failure? |
| 3 | Is IL-6 blockade safe in subjects with demyelinating disease? |
| 4 | What is the therapeutic effect of IL-6 blockade on structural joint damage in RA? |
| 5 | What are the consequences of IL-6 blockade on general mortality and that of cardiovascular cause in patients with RA? |
| 6 | Is there a relationship between IL-6 blockade and the induction of neoplasia (skin, solid organ, lymphomas) in patients with RA? |
| 7 | Is there any special clinical profile (in which its efficacy has been demonstrated as superior) or clinical manifestations of RA in which IL-6 blockade is particularly useful? |
| 8 | What is the risk of pulmonary involvement, deriving from IL-6 blockade, in subjects with RA? |
| 9 | Does therapeutic IL-6 blockade have any effect on the hydrocarbon metabolism of individuals with RA? |
| 10 | Which clinical profiles of children with JIA (systemic or polyarticular) could benefit from therapeutic IL-6 blockade, and to what extent (efficacy)? |
| 11 | What are the advantages (whether it is more effective), in a patient with RA and primary/secondary failure with an anti-TNFα of switching to therapeutic IL-6 blockade or trying a second anti-TNFα agent? |
| 12 | What is the evidence (efficacy and safety) for IL-6 blockade optimisation in RA? |
| 13 | Which is the best disease activity index to use in patients treated with IL-6? Is CRP the best disease activity index in RA with the use of anti-IL-6? |
| 14 | What is the efficacy of inhibition of IL-6 in RA in patients with RA after failure with synthetic DMARDs or MTX compared to other biologics or kinase inhibitors? |
| 15 | Do remission percentages increase in patients treated with IL-6 inhibitors due to their effect on CRP or ESR? |
| 16 | Do IL-6 inhibitors increase the risk of bowel perforation? |
| 17 | How do weight, smoking or the presence of RF and anti-CCP antibodies affect therapeutic response to IL-6 inhibition? |
| 18 | Are there differences between the different IL-6 inhibitors (efficacy, safety)? |
| 19 | How does IL-6 inhibition affect extra-articular manifestations of RA? |
| 20 | What is the efficacy and safety of IL-6 blockade in patients previously treated with non anti-TNFα biologics? |
| 21 | What is the effect of biological drugs or kinase inhibitors in patients who do not respond to IL-6 blockade? |
| 22 | Is one administration route safer and more effective than another? |
| 23 | What is the safest and most effective dose of an IL-6 inhibitor? |
JIA: juvenile idiopathic arthritis; anti-CCP: anti-citrullinated proteins; RA: rheumatoid arthritis; disease-modifying antirheumatic drugs; RF: rheumatoid factor; IL-6: interleukin-6; MTX: methotrexate; CPR: C reactive protein; TNFα: tumour necrosis factor α; ESR: erythrocyte sedimentation rate.
The group of experts held a nominal group meeting. Prior to the meeting, the result of the systematic review of the literature and a summary of the evidence found were sent in order to answer the questions posed by the coordinators. The recommendations and evidence were presented and discussed at the meeting.
This led to the formulation of the final recommendations, which were then dealt with through a Delphi vote (first round), using the televoting system, to assess the grade of agreement (GA). Agreement was defined if at least 80% of the participants answered yes to the recommendation (yes or no). The Delphi results were shown to the experts. In a second Delphi round, votes that reached a sufficient grade of agreement were again voted from 1 to 10 (1: totally disagree; 10: totally agree).
Final consensus documentThe final document was drawn up with all this information. Each of the recommendations, with the assistance of the methodologist, was assigned a level of evidence (LE) and a grade of recommendation (GR) according to the recommendations of the Oxford Centre for Evidence-Based Medicine.28 Allocation of GA was as described. The paper was circulated among the experts for final appraisal and final comments.
ResultsTwo IL-6 inhibitors have currently been approved in our country: TCZ (marketed) and SAR (in the process of marketing), whose main characteristics are shown in Table 2.
Main characteristics of tocilizumab and sarilumab.
| Characteristic | Tocilizumab | Sarilumab |
|---|---|---|
| Molecule | Recombinant human IgG monoclonal antibody | Recombinant human IgG monoclonal antibody |
| Mechanism of action | Blockade of both soluble IL-6 and membrane-bound receptors | Blockade of both soluble IL-6 and membrane-bound receptors |
| Administration route | IV, SC | SC |
| Steady-state half life | 12 days | 21 days |
| Treatment dose | IV formulation 8mg/kg every 4 weeksSC formulation 162mg every week | SC formulation 200mg every 2 weeks |
| Presentation | Infusion vialPreloaded syringe | Pre-filled syringe and pen |
| Indication on technical datasheet | -In combination with MTX for:• Severe, active and progressive RA in adults not treated previously with MTX• Active, moderate-severe RA in adult patients who have not responded appropriately, or who are intolerant to ≥1 DMARD or TNF antagonists-TCZ can be administered as monotherapy if there is intolerance to MTX or when continuous treatment with MTX is inappropriate | -In combination with MTX for:• Active, moderate-severe RA in adult patients who have not responded appropriately, or who are intolerant to ≥1 DMARD-SAR can be administered in monotherapy in the case of intolerance to MTX or when treatment with MTX is not appropriate |
RA: rheumatoid arthritis; DMARDs: disease-modifying antirheumatic drugs; IV: intravenous; kg: kilogramme; mg: milligram; MTX: methotrexate; SC: subcutaneous; TNF: tumour necrosis factor.
TCZ has been available to clinical rheumatologists since 2009, and both its intravenous (iv) and subcutaneous (sc) formulations are available. There is therefore a large amount of both short-term and long-term information from RCTs and observational studies. They have been studied in early RA, established RA, RA naïve to traditional or biological disease modifying drugs, refractory/intolerant RA, in monotherapy, and in combination.18,29–38 We also have data on the effect of TCZ in monotherapy once its use has been discontinued,39 and on its efficacy when reintroduced.40 It has been shown to achieve clinical remission or low activity in a significant percentage of patients.18,29,30,32,33,38 It can be observed at 2 weeks, and can last up to several years.32,33,36,41 TCZ IV in monotherapy is superior to adalimumab,29 methotrexate (MTX)34,35 and other traditional DMARDs42 in monotherapy. When combined with another DMARDs, the percentages of remission increases, although this is not statistically significant.32,33,36,41 Radiological progression was analysed in 5 articles,31,43 with plain radiography or magnetic resonance imaging.31 TCZ IV in monotherapy delays radiological progression at least to one year of treatment. This effect is greater for RA with a greater risk of unfavourable outcome.43 The efficacy of TCZ in RA has also been demonstrated in other variables such as functional capacity,30,32–35,41,42,44,45 bone metabolism,30,44 fatigue, C reactive protein (CRP), quality of life, pain, morning stiffness and anaemia.18,29,32,33,35–38,41,44,45
SAR9,46–49 was studied in established cases of RA refractory/intolerant to MTX, other traditional DMARDs and TNF inhibitors, in monotherapy or in combination with MTX. SAR (200mg every 2 weeks) achieves clinical remission or low activity in a significant percentage of patients; it improves functional capacity and other patient- related outcomes (PRO), and reduces radiographic progression for up to 52 weeks of treatment. There is also intermediate data showing maintained efficacy at two years.49 Like TCZ, its efficacy in monotherapy was superior to adalimumab in monotherapy for most of the variables studied.10 Finally, other variables studied with a good response to SAR were, for example, fatigue, CRP, anaemia, and fasting blood sugar and glycated haemoglobin.9,46–49
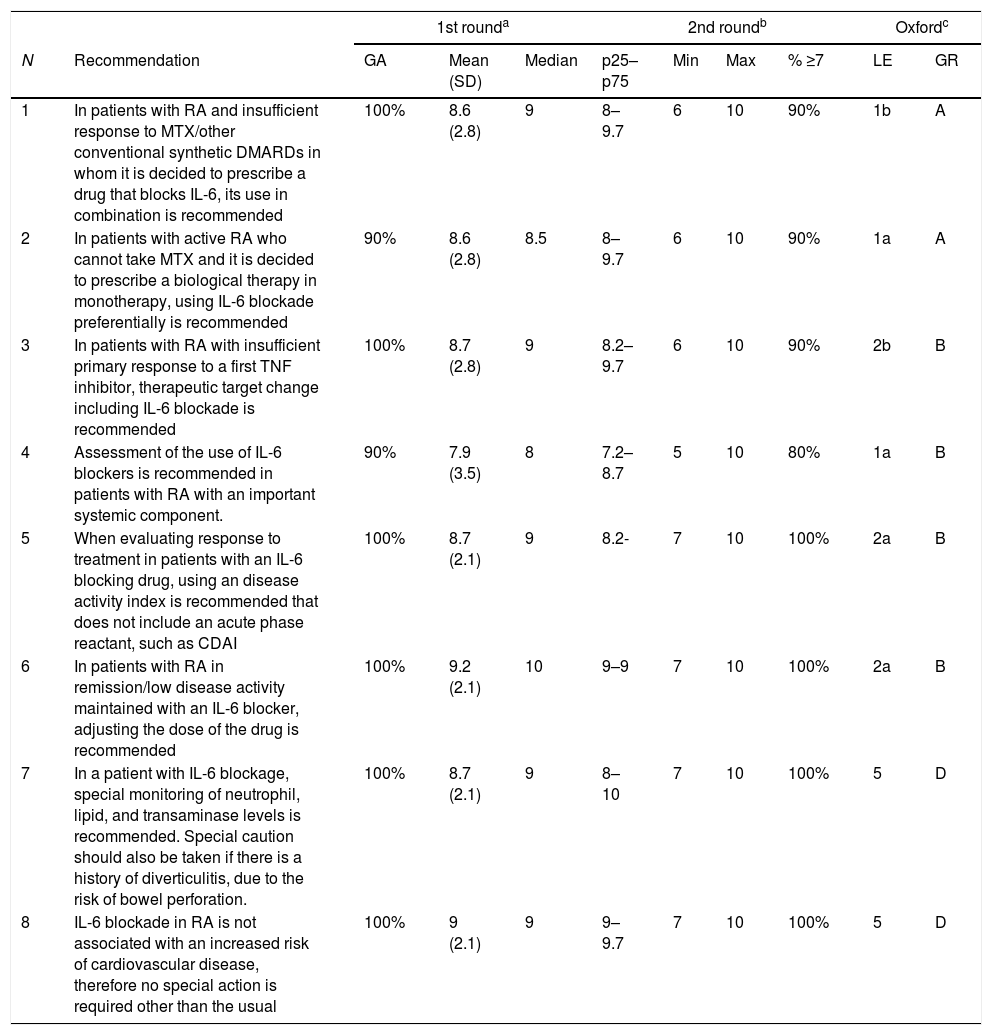
Below, we discuss the recommendations and full evidence with the use of IL-6 inhibitors (see Table 3 for the full Delphi results).
Delphi technique (1st and 2nd round) results of the recommendations on the use of IL-6 inhibitors in patients with RA.
| 1st rounda | 2nd roundb | Oxfordc | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | Recommendation | GA | Mean (SD) | Median | p25–p75 | Min | Max | % ≥7 | LE | GR |
| 1 | In patients with RA and insufficient response to MTX/other conventional synthetic DMARDs in whom it is decided to prescribe a drug that blocks IL-6, its use in combination is recommended | 100% | 8.6 (2.8) | 9 | 8–9.7 | 6 | 10 | 90% | 1b | A |
| 2 | In patients with active RA who cannot take MTX and it is decided to prescribe a biological therapy in monotherapy, using IL-6 blockade preferentially is recommended | 90% | 8.6 (2.8) | 8.5 | 8–9.7 | 6 | 10 | 90% | 1a | A |
| 3 | In patients with RA with insufficient primary response to a first TNF inhibitor, therapeutic target change including IL-6 blockade is recommended | 100% | 8.7 (2.8) | 9 | 8.2–9.7 | 6 | 10 | 90% | 2b | B |
| 4 | Assessment of the use of IL-6 blockers is recommended in patients with RA with an important systemic component. | 90% | 7.9 (3.5) | 8 | 7.2–8.7 | 5 | 10 | 80% | 1a | B |
| 5 | When evaluating response to treatment in patients with an IL-6 blocking drug, using an disease activity index is recommended that does not include an acute phase reactant, such as CDAI | 100% | 8.7 (2.1) | 9 | 8.2- | 7 | 10 | 100% | 2a | B |
| 6 | In patients with RA in remission/low disease activity maintained with an IL-6 blocker, adjusting the dose of the drug is recommended | 100% | 9.2 (2.1) | 10 | 9–9 | 7 | 10 | 100% | 2a | B |
| 7 | In a patient with IL-6 blockage, special monitoring of neutrophil, lipid, and transaminase levels is recommended. Special caution should also be taken if there is a history of diverticulitis, due to the risk of bowel perforation. | 100% | 8.7 (2.1) | 9 | 8–10 | 7 | 10 | 100% | 5 | D |
| 8 | IL-6 blockade in RA is not associated with an increased risk of cardiovascular disease, therefore no special action is required other than the usual | 100% | 9 (2.1) | 9 | 9–9.7 | 7 | 10 | 100% | 5 | D |
RA: rheumatoid arthritis; CDAI: Clinical Disease Activity Index; SD: standard deviation; DMARDs: disease-modifying antirheumatic drugs; GA: grade of agreement; GR: grade of recommendation; IL-6: interleukin-6; Max: maximum; Min: minimum; MTX: methotrexate; LE: level of evidence; p25–p75: percentiles 25 and 75; TNF: tumour necrosis factor.
Recommendation 1: In patients with RA and insufficient response to conventional synthetic MTX/other DMARDs, for whom it is decided to prescribe a drug that blocks IL-6, use in combination is recommended (LE1b; GR A; GA 90%).
The panel first wants to stress that it is important that this option is individualised for each case assessing the balance between risk (toxicity) and benefit (magnitude of effect, if clinically relevant). Also, most of the evidence comes from combining with MTX, although we also have evidence (far less) with leflunomide, salazopyrine and antimalarials.15,50
For TCZ, we have many RCT in which, in terms of activity or harm, no statistically significant differences were generally found between monotherapy and combination with MTX or DMARDs. However the trend and absolute numbers favour combination, especially with prominent variables such as ACR70, longer-term radiological damage or for patients who are refractory to TNF inhibitors.51–54,43,55
With respect to SAR, it has been published that, in patients with established RA refractory to MTX, the combined use of SAR and MTX is associated with a greater reduction in the Sharp/van der Heijde index at 52 weeks compared to the MTX in monotherapy group.12
Recommendation 2: In patients with active RA who cannot take MTX and those for whom prescription of a biological drug in monotherapy has been decided, it is recommended that agents that block IL-6 should be used preferentially (LE1a; GR A; GA 90%).
It should be noted that the criteria for not prescribing MTX would be contraindication, intolerance or medical judgement. Most of the evidence refers to patients who cannot take specific MTX, although this could probably be extended to other synthetic DMARDs.
The panel wishes to point out that there are other therapeutic options in addition to IL-6 blockade for these types of patients, an indication included in its technical data sheets, but for which there is less evidence regarding its use in monotherapy.56–58
In terms of the evidence, in patients with established, active RA who are intolerant or for whom it is inappropriate to continue with MTX and compared with adalimumab,29,59 TCZ IV was superior (at 24 weeks) in variables such as clinical remission, low disease activity or Clinical Disease Activity Index (CDAI). On the other hand, no significant differences were observed between adalimumab and TCZ in improvement in the Health Assessment Questionnaire (HAQ), fatigue, or the health-related quality of life questionnaire (SF 36). In a similar patient profile, which also included patients refractory to MTX10, SAR was superior (at 24 weeks) in clinical remission, CDAI remission and low disease activity, at week 12 and at week 24, with statistical significance, the HAQ and SF-36 (physical component). However, there was no significant difference between adalimumab and SAR in the improvement of fatigue and the SF-36 mental component. There are preliminary data with other IL-6 blockers that point in the same direction.21,23
Recommendation 3: In patients with RA with insufficient primary response to a first TNF inhibitor, a change of therapeutic target is recommended, including IL-6 blockade (LE 2b; GR B; GA 90%).
It has been demonstrated in the literature that TCZ is effective in patients refractory to TNF30,35–37 both in the short60 and long term.61 The efficacy of SAR (along with a conventional DMARD) has also been analysed in these patients, at least in the short term.11 SAR was superior to placebo in both disease activity and response rates (DAS 28-PCR, ACR 20/50/70), as well as functional capacity (HAQ)11 and patient-perceived health variables, including work productivity.47 There is some short-term (12 weeks) data for olokizumab on its superiority to placebo in these patients.21,22 We also have similar data for sirukumab,24 although this drug is not going to be marketed for the time being.
The panel also considers that, in cases of inefficacy secondary to TNF inhibitor, the decision to change therapeutic target is not as clear, and is not supported by the evidence. Therefore, for example, in cases where this is due to immunogenicity, another TNF inhibitor can be tested. However, this does not preclude a change of TNF inhibitor target.
In cases of class adverse events, such as demyelinating disease with TNF inhibitors, the panel also recommends a change of therapeutic target.
Recommendation 4: It is recommended that the use of IL-6 blockade in patients with RA with a significant systemic component should be assessed (LE 1a; GR A; GA 80).
The panel defined a clinically important systemic component (clinically significant anaemia due to disease, fatigue, and fever/slight fever).
The literature confirmed that patients with anaemia had a significant increase in haemoglobin levels with TCZ compared to those treated with MTX (1.19 vs .10g/dL),35 whereas in other studies the increase was 6–7g/L compared to placebo.51 There are similar results with other IL-6 inhibitors such as SAR.62 There are also data on the efficacy of IL-6 inhibitors in fatigue,15,22,36,63,64,51,65,66 pain,15,46,51,65–68 morning stiffness,15,66 and depression.64,66
On the other hand, with regard to other clinical profiles such as patients with amyloidosis, case series of patients with this manifestation under treatment with TCZ have been published and no problems recorded.69 In patients with lung disease, the use of an IL-6 blocking agent has not been clinically shown to cause a specific lung disease, nor has it been shown to be associated with the development or exacerbation of a lung disease.70–73 Finally, in terms of bone metabolism, there are data indicating that TCZ in monotherapy,30,44 and SAR in monotherapy and combined therapy may have a beneficial effect in this regard.9,74
Little information is currently available on the effect of these drugs on hydrocarbon metabolism. There is a small observational study with TCZ, where it appears that the drug could improve insulin sensitivity (decrease resistance).75 It has also been seen in RCT subanalysis of SAR48,76 that, in patients with RA with or without diabetes, the use of SAR with traditional DMARDs decreased fasting blood sugar and glycated haemoglobin levels. The decrease in glycated haemoglobin in diabetic patients compared to placebo was very significant.
Recommendation 5: When evaluating response to treatment in patients with an IL-6 blocking drug, use of a disease activity index is recommended that does not include an acute-phase reactant, such as CDAI (LE 2b; GR B; GA 100%).
IL-6 blockade has been shown to directly and rapidly inhibit the production of acute phase reactants, especially CRP.13,22,25,50,77 Therefore, CRP and the indices containing this reactant may not be good indices for monitoring the efficacy of these drugs. Thus, the European consensus document on IL-6 blockade and related documents78 propose the use of CDAI as a compound index to monitor disease activity. This same document also considers better indices that use erythrocyte sedimentation rate instead of CRP.79
Recommendation 6: In patients with RA in remission/low activity with IL-6 blockade, the panel recommends assessing adjustment of the drug dose (LE 2a; GR B; GA 100%).
First, the panel considered that the patient should have been in remission/low disease activity on a sustained basis (6 months) to consider dose adjustment.
Several systematic reviews of the literature with TCZ (principally based on observational studies), including its used as a second line biologic, show that dose adjustment in patients in remission is reasonable.80,81 Similarly, a retrospective Spanish observational study maintains that optimisation in patients in remission is effective even when a change is made from IV to SC.82 Recently in an RCT, patients who were in sustained remission after 24 weeks of treatment with 162mg of TCZ SC weekly (in monotherapy and in combination) were randomised to either continue this regimen or change to every 2 weeks. It was observed that after a further 24 weeks, the percentage of patients in clinical remission was 73.9% in those whose doses were adjusted.83 We should point out that in the United States the approved dose of TCZ is 4mg, based on RCTs in which this dose was effective in a significant percentage of patients.51,41 In the open phase of RCTs with SAR, the dose was reduced from 200mg to 150mg if there were laboratory abnormalities (neutropenia, thrombocytopenia or elevated transaminases), not for reasons of optimisation. On the other hand, an analysis found that this occurred in 17% of patients, in which, in addition to improving safety parameters, efficacy was maintained in the majority at 24 weeks.84
Finally, no dose regimen adjustment is better than another (increasing the interval, reducing the dose). Different ways of doing this have been published, therefore the panel did not opt for any one in particular.85 Some of the most described are, for TCZ IV, a reduction in dose to 4mg or 6mg maintaining the interval and for TCZ SC, increasing the interval to 14 days.
The panel would like to comment that, in relation to other biological drugs, risk management in general is carried out in the same way. We shall go on to review this management and outline specific peculiarities that we found in the use of these drugs.
Recommendation 7: In a patient with IL-blockage, monitoring of neutrophil, lipid and transaminase levels especially is recommended. Special care is required if the patient has a history of diverticulitis, due to the risk of bowel perforation. (LE 5; GR D; GA 100%).
With the use of IL-6 inhibitors we can observe a marked decrease in neutrophils already in the first 4 weeks of treatment.10,25,47,51,86,87 However, clinically significant neutropenia occurs in ∼3% of patients and is usually transient.12,13,86 In most cases, neutrophil counts return to normal even when the drug is continued. But most important in this regard is that there is no association between neutropenia and the onset or intensity of infections.10,25,47,51,86,87 Furthermore, the rate of serious infections is similar to that observed with other biologics such as TNF inhibitors.88
For monitoring purposes, the panel recommends following the instructions given on the technical data sheet of these drugs and the recommendations of the SER.89 And in the case of recurrent or major (not clinically significant) neutropenia, shortening the monitoring time will be assessed based on clinical judgement.
With regard to clinical monitoring, it is important to bear in mind that some symptoms or general signs of infection may not be present or may be masked by IL-6 blockade (acute phase reactants, fever or leucocytosis),90 therefore, we need to be clinically cautious.
IL-6 inhibitors produce increases in the levels of the different serum lipids, but the overall atherogenic quotient is unchanged and there has been no evidence of an increase in cardiovascular events (even with long-term use).11,17,18,21,23,24,47,91,92 Lipids increase in the first few weeks after starting treatment and their levels remain stable over time. It should be noted that patients with hyperlipidaemia due to these drugs have an adequate response to statins.50 The panel advises following the usual recommendations for hypercholesterolaemia.93,94
An increase in transaminases is relatively frequent with IL-6 inhibitors, although the majority (≥90%) are mild (<3 LSN) and transient (even without suspension),7,10–12,18,23,25,47 and are easily managed following the usual monitoring recommendations.89 Serious cases that are similar to those found in patients with RA treated with other biologics and traditional DMARDs are exceptional.95 The elevation of transaminases is clearly higher in patients treated with TCZ in combination with MTX.95,96
On the other hand, in studies published with TCZ,7,97–100 the rate of gastrointestinal perforation is very low 2.2/1000 patients/year in the SAR studies.10–12 Both have been associated with a history of diverticulitis, concomitant use of NSAIDs and corticosteroids, as well as a higher number of previous biological drugs.10,99,100 This risk is slightly increased compared to that found for the traditional DMARDs or TNF inhibitors, and is less than of corticosteroids.100
Again, we must bear in mind that, due to the effects of IL-6 blockade, the clinical symptoms of diverticulitis can be masked.101,102
Furthermore, we should point out that thrombocytopenia is rare and, from a clinical point of view, is usually of little relevance (see the technical datasheets for monitoring of thrombocytopenia).101,102
Recommendation 8: IL-6 blockage in RA is not associated with an increased risk of cardiovascular disease, and therefore no special action other than the usual are required (LE 5; GR D; GA 100%).
In terms of general mortality rate, there are no data to indicate that this increases with any drug that blocks IL-6, or that it is different to that recorded with other biological therapies.24,103,104 Therefore, in relation to cardiovascular mortality, different RCT and observational studies have shown that TCZ does not increase the incidence of cardiovascular disease (acute myocardial infarction [AMI] or acute cerebrovascular accident [ACVA]) either in the short or long term, and this is not associated with time of exposure or route of administration and dose, and that the rates recorded are similar to those expected in the population of patients with RA.7,18,38,86 On the other hand, with regard to SAR, in the published RCTs (up to 52 weeks) only one case of ACVA resulting in death has been described with SAR 200mg, in a total of almost 2800 patients.62 We have little evidence for the rest of the drugs.
The panel advises following the recommendations of the principal scientific societies on cardiovascular risk prevention and management.93,94
In RCTs and their extensions (short and long term), and in observational studies (close, or routine clinical practice) on TCZ103,105 heart failure has not been reported as either a frequent adverse event or one associated with use of the drug. Similarly, in short and medium term RCTs with SAR,10,62,106–109 it has not been found to be a frequent adverse event or one associated with use of the drug. Finally, with regard to the remaining IL-6 inhibitors, in short- to medium-term (12–54 weeks depending on the drug) RCTs of olokizumab,21,22 sirukumab,25,110,111 and clazakizumab,23 the results have been similar.
The TCZ datasheet,101 in the section on special warnings and precautions for use, states the following: “Physicians should be vigilant for symptoms potentially indicative of new-onset central demyelinating disorders. The potential for central demyelination with TCZ is currently unknown”. The datasheet for SAR does not indicate anything in this regard.102
In RCTs, in extension studies (short and long term) and in observational studies (close or routine clinical practice), there are no events that indicate that the use of TCZ is associated with demylenating disorders.7 There are isolated cases of patients with RA and multiple sclerosis who have been prescribed TCZ for RA without it having worsened their multiple sclerosis.112 At present there is no data relating to other IL-6 inhibitors. On the other hand, there are observational studies where patients with demyelinating diseases, such as neuromyelitis optica, have been treated with TCZ with good results.113
Regarding cancer, with the use of IL-6 inhibitors, TCZ has not been associated with greater development of neoplasia86,114–116 and the rate found is comparable to that detected in patients under treatment with synthetic DMARDs, and does not increase with exposure time or administration route or dose. The data for SAR,62 point in the same direction.
The panel advises following the recommendations on the technical datasheets and consensuses on risk management when using these therapies.89,101,102
Finally, to date it has not been clinically demonstrated that the use of IL-6 blockade causes a specific lung disease in subjects with RA. The use of TCZ has not been associated with the development or exacerbation of any lung disease, such as interstitial lung disease (in either RCTs or observational studies).70–73 Although there is insufficient data, the rest of the IL-6 inhibitors point in the same direction.
DiscussionWe present a series of recommendations in this paper on the use of IL-6 inhibitors in RA, focussing on the aspects considered to contribute most in daily practice, based on the best available evidence. In addition, we synthesised all the evidence gathered, in terms of efficacy and risk management.
For this purpose we followed the nominal group and Delphi techniques, widely used in these types of documents.89 We also selected a group of nationally recognised experts, supported by a systematic review with the help of an expert methodologist.
The important changes that have appeared in recent years in the treatment of RA1–4 are well known. One of these changes has been the incorporation of the biological therapies. These were initially the TNF inhibitors, after which drugs with different mechanisms of action have been emerging, which include those that block IL-6, both the receptor and the cytokine itself, although only the former have been approved for use in RA (TCZ and SAR).6–18
In this context, national and international organisations are trying to issue and update recommendations on the treatment of RA.1,117 However, bearing in mind that we have a very wide therapeutic arsenal and a great deal of scientific evidence, it is not always possible to delve deeper into the fine management of these therapies. For this reason, there are also documents by experts that focus on the use of a specific therapeutic target. A document was published 5 years ago on the use of IL-6 inhibitors.79
However, since then new drugs have emerged and there is much new evidence, which justifies a new recommendation document on these drugs, as in this paper. In addition, and to avoid repetition, the panel decided to address the most clinically relevant aspects of the use of these therapies.
Thus, from the point of view of efficacy and indication, we approached the use of these drugs in monotherapy and combined therapy, and in patient profiles of clinical interest, such as those for whom TNF inhibitors have failed, and those with an important systemic component. The latter, due to the potent effect at this level of these drugs’ mechanism of action. This systemic effect also includes a very marked decrease in acute phase reactants, usually included in many compound disease activity indices, which has led to specific recommendations on how to assess treatment response more accurately and reliably.
Also, because of its mechanism of action, the panel considered it clinically important to be aware of certain adverse events. Although many of them are known and therefore monitoring is similar to that of other biological therapies, the panel considered it very important to emphasise those that are characteristic of IL-6 blockade such as neutropenia, risk of bowel perforation, dyslipidaemia and cardiovascular prevention.
Furthermore, although the systematic review of the literature was very extensive, the main limitation is the difficulty in establishing very specific recommendations in the different lines of treatment in relation to other biological therapies, since there are practically no data in this regard. Nor do we currently have studies comparing the different IL-6 inhibitors.
In summary, in order to improve clinical practice, it is essential to have explicit recommendations on the role of IL-6 inhibitors in RA. Although evidence is still scarce in some areas, this document presents a number of recommendations that we believe could be relevant and useful for clinicians. In addition, everyday aspects related to their use are referred to, which means that the recommendations can be implemented without difficulties in daily practice. The panel is convinced that monitoring will contribute to more rational use of the drug, with the consequent improvement in management, prognosis and quality of life.
FinancingSanofi Spain provided funds to Inmusc to support the systematic review and development of the recommendations, but did not intervene in the project design or development of the recommendations, they only undertook a courtesy review.
Conflict of interestsHC reports having received honoraria for talks, collaborations and courses from Sanofi, Lilly, MSD, Abbvie, Gebro. JTM has received funds for research or training from Gebro Pharma, Janssen, Lilly, Novartis, Pfizer and Sanofi. The remaining authors have no conflicts of interest to declare.
Please cite this article as: Tornero Molina J, Balsa Criado A, Blanco García F, Blanco Alonso R, Bustabad S, Calvo Alen J, et al. Recomendaciones de experto sobre el bloqueo de la interleucina 6 en pacientes con artritis reumatoide. Reumatol Clin. 2020;16:272–281.