To determine positivity and clinical correlation of anti-neutrophil cytoplasmic antibodies (ANCA), taking into account the interference of antinuclear antibodies (ANA).
Material and methodsA prospective study was conducted in the Laboratory of Immunology of the National Cuban Center of Medical Genetic during one year. Two hounded sixty-seven patients with indication for ANCA determination were included. ANCA and ANA determinations with different cut off points and assays were determined by indirect immunofluorescense. Anti proteinase 3 and antimyeloperoxidase antibodies were determined by ELISA.
ResultsMost positivity for ANCA was seen in patients with ANCA associated, primary small-vessel vasculitides, rheumatoid arthritis and systemic lupus erythematosus. Presence of ANCA without positivity for proteinase 3 and myeloperoxidase was higher in patients with ANA and little relation was observed between the perinuclear pattern confirmed in formalin and specificity by myeloperoxidase. Highest sensibility and specificity values for vasculitides diagnostic were achieved by ANCA determination using indirect immunofluorescense with a cut off 1/80 and confirming antigenic specificities with ELISA.
ConclusionANCA can be present in a great number of chronic inflammatory or autoimmune disorders in the population studied. This determination using indirect immunofluorescence and following by ELISA had a great value for vasculitis diagnosis. Anti mieloperoxidasa assay has a higher utility than the formalin assay when ANA is present.
Determinar la positividad y la correlación clínica de los anticuerpos contra el citoplasma del neutrófilo (ANCA), teniendo en cuenta la interferencia de los anticuerpos antinucleares (ANA).
Material y métodoSe realizó un estudio prospectivo en el Laboratorio de Inmunología del Centro Nacional de Genética Médica de Cuba durante un año. Se incluyó a 267 pacientes con indicación de ANCA. Las determinaciones de ANCA a diferentes puntos de corte y de ANA se realizaron mediante inmunofluorescencia indirecta. Los anticuerpos antiproteinasa 3 y antimieloperoxidasa fueron determinados mediante ELISA.
ResultadosNuestro estudio mostró que la mayor positividad de ANCA fue vista en pacientes con vasculitis asociadas a ANCA, artritis reumatoidea y lupus eritematoso sistémico. Fue superior la presencia de ANCA sin especificidad por la proteinasa 3 o la mieloperoxidasa en pacientes con ANA y se observó poca relación entre el patrón perinuclear confirmado en formalina y la presencia de anticuerpos frente a la mieloperoxidasa. Los mayores valores de sensibilidad y especificidad para el diagnóstico de las vasculitis se alcanzaron para la determinación de ANCA mediante inmunofluorescencia indirecta a un valor de corte de 1/80 y confirmando la especificidad antigénica mediante ELISA.
ConclusionesLos ANCA pueden estar presentes en un amplio número de enfermedades asociadas a estados inflamatorios y autoinmunes en la población estudiada. Su determinación mediante inmunofluorescencia indirecta seguida de la determinación mediante ELISA tiene gran valor para el diagnóstico de las vasculitis. La determinación de anticuerpos antimieloperoxidasa tiene mayor utilidad que el ensayo en láminas de formalina cuando hay ANA.
Anti-neutrophil cytoplasm antibodies (ANCA) are a group of autoantibodies directed against the cytoplasmic constituents of neutrophils and monocytes. Their determination constitutes a diagnostic test for small-vessel vasculitis associated with ANCA (AAV) including granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA), eosinophilic granulomatosis with polyangiitis (EGPA) and vasculitis limited to the kidney.1
The presence of ANCA is determined by indirect immunofluorescence (IIF) from fixed neutrophils on glass slides and fluorescence positivity must be confirmed by the enzyme-linked immunosorbent assay enzyme (ELISA) to determine the antigenic specificity, although some authors suggest that the simultaneous use of both methods has shown greater diagnostic value.2
There are two main patterns of fluorescence depending on the purpose of the ANCA. The cytoplasmic pattern (cANCA) refers to the pattern with which the cytoplasm stains under immunofluorescent microscope when neutrophils are fixed with ethanol or acetone. The main cANCA antigen is proteinase 3 (PR3). The perinuclear pattern (pANCA) refers to the staining pattern of neutrophils, which occurs only when they are fixed in ethanol or acetone, to permeabilize the membrane of the cytoplasmic granules, and allows highly cationic proteins, such as myeloperoxidase (MPO), to exit and bind to the negatively charged nuclear membrane. When neutrophils are fixed in formalin, this pANCA pattern is observed as cANCA because the attraction effect of cationic proteins to the nucleus1–3 is reduced.
Over 90% of patients with active typical GPA have identifiable antibodies against PR3, with a sensitivity of 28%–92%, depending on the expression of the disease, and a specificity of 80%–100%; while between 80% and 70% of patients with PAM and between 70% and 85% of EGPA patients have identifiable MPO antibodies.4,5
It has been confirmed that the pANCA pattern can be observed when the antibodies react with antigens other than MPO, something called an atypical pANCA pattern. Perinuclear fluorescence without nuclear extension is also known as atypical pANCA.6,7
Some authors have also suggested that there is a cANCA atypical pattern combining perinuclear fluorescence with nuclear extension and cytoplasmic fluorescence. This pattern can be produced by the interference of antinuclear antibodies (ANA).6,7
Hence the importance of the simultaneous determination of ANA and ANCA determination, although there are few studies on the prevalence of ANCA patterns by IIF in different diseases, differentiating the presence or absence of ANA.
Atypical patterns may have antigenic specificities against nuclear, cytosolic or granular components and they include elastase, cathepsin G, bactericidal permeability enhancer (BPE), betaglucoronidase, lysozyme, lactoferrin, catalase, alpha enolase, actin, histone, among others.7,8
Some authors suggest the usefulness of ANCA is not only to diagnose AAV, but also to diagnose and assess the prognosis of other inflammatory or autoimmune disorders, where the pattern of associated IIF is usually described as pANCA, although there have been cANCA or atypical patterns.1,7,9,10 ANCA positivity has also been described in infectious and malignant hematological diseases.1,11,12
The objective of this study was to determine the positivity and clinical correlation of ANCA, taking into account the interference of ANA in patients treated at the Laboratory of Immunology at the National Centre for Medical Genetics (NCMG) of Cuba in 2012.
Materials and MethodsA prospective study was performed with patients referred to the Laboratory of Immunology of the NCMG of Cuba in 2012.
PatientsThe sample consisted of 267 patients with indications for ANCA determination: 35 with suspected systemic lupus erythematosus (SLE), 60 with confirmed SLE, 17 with suspected AAV,13 with AAV, 91 with rheumatoid arthritis (RA), 10 with viral hepatitis, 14 with scleroderma, 8 with mixed connective tissue disease, 4 with Sjögren's syndrome, five with autoimmune hepatitis (AIH) type i, 3 with ulcerative colitis (UC), 3 with Raynaud's phenomenon, 2 with dermatomyositis, one with primary biliary cirrhosis and one with spondyloarthropathy. The diagnosis of AAV was performed according to the criteria established by the Conference of Chapell Hill.13–15 The diagnosis of other patients was performed by clinicians and immunologists with criteria for each disease.
MethodsANA determination was performed by IIF, considering as positivity titers starting at 1/80 and using a commercial assay (ORGENTEC, Germany). The determination of ANCA was performed by IIF on ethanol-fixed and formalin human neutrophils, considering positivity from 1/2016; determinations of anti-PR3 and anti-MPO were performed by ELISA (ORGENTEC, Germany) to patients who were positive for ANCA by IIF, using as cutoff 5IU / mL. 2 IIF patterns were identified: cANCA (cytoplasmic fluorescence), pANCA (perinuclear fluorescence without nuclear extension).ANCA positive samples were titrated by IIF. The titer was considered as the highest dilution at which the ANCA pattern was clearly observed. Sensitivity and specificity of ANCA determination by IIF was determined using different cutoffs and confirming the antigenic specificities by ELISA.
For statistical analysis we used the Statistica 7.0 and 3.1 EPIDAT programs. To analyze the influence of ANA positivity in determining ANCA, the ¿2 statistic was calculated as well as odds ratios (OR) and the magnitude of association with their respective confidence interval of 95%. Different cutoffs were compared in terms of their discrimination capability, using the area under the curve (AUC) of the receiver operating curve characteristics (ROC curves).
Research fulfilled the Declaration of Helsinki of the World Medical Association criteria, which establishes the ethical principles for medical research involving human subjects.17 The study was approved by the Ethics Committee of the NCMG.
Informed consent for participation was obtained from patients in the office, with sufficient time to decide whether to participate and before blood was drawn.
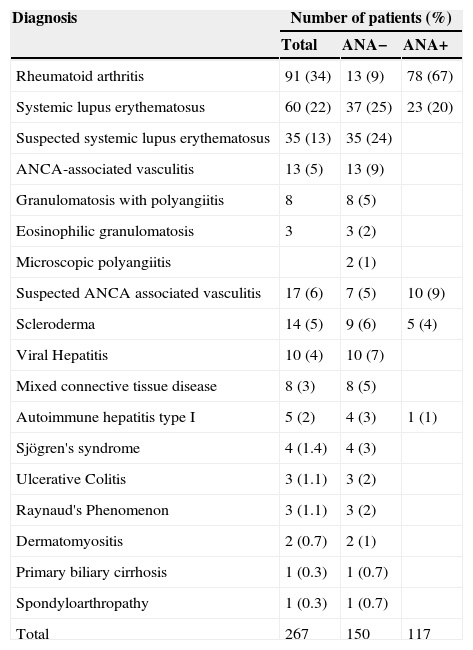
ResultsThe mean age of the patients included in the study was 40 years, ranging from 1 to 75 years. Only 17% of patients had an age greater than or equal to 60 years. 73% of patients (195) were females and of the total group of patients, 56% (150) were negative for ANA (Table 1).
Number of Patients Included in the Study With Positive Antinuclear Antibody.
| Diagnosis | Number of patients (%) | ||
|---|---|---|---|
| Total | ANA− | ANA+ | |
| Rheumatoid arthritis | 91 (34) | 13 (9) | 78 (67) |
| Systemic lupus erythematosus | 60 (22) | 37 (25) | 23 (20) |
| Suspected systemic lupus erythematosus | 35 (13) | 35 (24) | |
| ANCA-associated vasculitis | 13 (5) | 13 (9) | |
| Granulomatosis with polyangiitis | 8 | 8 (5) | |
| Eosinophilic granulomatosis | 3 | 3 (2) | |
| Microscopic polyangiitis | 2 (1) | ||
| Suspected ANCA associated vasculitis | 17 (6) | 7 (5) | 10 (9) |
| Scleroderma | 14 (5) | 9 (6) | 5 (4) |
| Viral Hepatitis | 10 (4) | 10 (7) | |
| Mixed connective tissue disease | 8 (3) | 8 (5) | |
| Autoimmune hepatitis type I | 5 (2) | 4 (3) | 1 (1) |
| Sjögren's syndrome | 4 (1.4) | 4 (3) | |
| Ulcerative Colitis | 3 (1.1) | 3 (2) | |
| Raynaud's Phenomenon | 3 (1.1) | 3 (2) | |
| Dermatomyositis | 2 (0.7) | 2 (1) | |
| Primary biliary cirrhosis | 1 (0.3) | 1 (0.7) | |
| Spondyloarthropathy | 1 (0.3) | 1 (0.7) | |
| Total | 267 | 150 | 117 |
ANA−: absence of antinuclear antibodies; ANA: antinuclear antibodies; ANCA: antibodies against neutrophil cytoplasm.
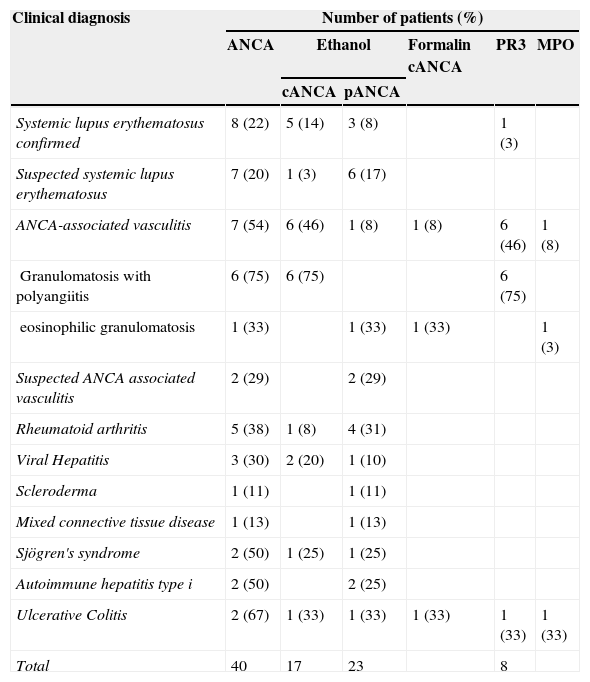
Of the 150 ANA negative patients, 40 (27%) had ANCA by IIF and of these 10 patients (25%) had anti-PR3 and anti-MPO (Table 2) antibodies. In this group of patients, the anti-PR3 antibodies corresponded to a cANCA pattern and all anti-MPO antibodies corresponded to a positive pANCA in formalin.
Positivity of Antibodies Against Neutrophil Cytoplasm Considering the Clinical Diagnosis of Patients Without Antinuclear Antibodies.
| Clinical diagnosis | Number of patients (%) | |||||
|---|---|---|---|---|---|---|
| ANCA | Ethanol | Formalin cANCA | PR3 | MPO | ||
| cANCA | pANCA | |||||
| Systemic lupus erythematosus confirmed | 8 (22) | 5 (14) | 3 (8) | 1 (3) | ||
| Suspected systemic lupus erythematosus | 7 (20) | 1 (3) | 6 (17) | |||
| ANCA-associated vasculitis | 7 (54) | 6 (46) | 1 (8) | 1 (8) | 6 (46) | 1 (8) |
| Granulomatosis with polyangiitis | 6 (75) | 6 (75) | 6 (75) | |||
| eosinophilic granulomatosis | 1 (33) | 1 (33) | 1 (33) | 1 (3) | ||
| Suspected ANCA associated vasculitis | 2 (29) | 2 (29) | ||||
| Rheumatoid arthritis | 5 (38) | 1 (8) | 4 (31) | |||
| Viral Hepatitis | 3 (30) | 2 (20) | 1 (10) | |||
| Scleroderma | 1 (11) | 1 (11) | ||||
| Mixed connective tissue disease | 1 (13) | 1 (13) | ||||
| Sjögren's syndrome | 2 (50) | 1 (25) | 1 (25) | |||
| Autoimmune hepatitis type i | 2 (50) | 2 (25) | ||||
| Ulcerative Colitis | 2 (67) | 1 (33) | 1 (33) | 1 (33) | 1 (33) | 1 (33) |
| Total | 40 | 17 | 23 | 8 | ||
ANCA: antineutrophil cytoplasmic antibodies; cANCA: cytoplasmic ANCA pattern; MPO: myeloperoxidase; pANCA: perinuclear ANCA pattern; PR3: proteinase 3.
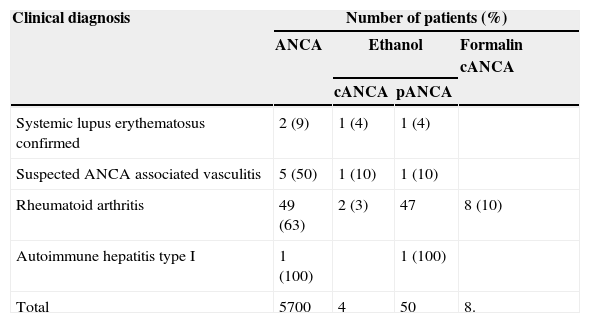
Of the 117 ANA positive patients, 57 (49%) had ANCA by IIF. All these positive ANCA patients by IIF were negative for anti-PR3 and anti-MPO antibodies. 51 pANCA patterns obtained, 6 were positive in formalin and were not associated with anti-MPO (Table 3) antibodies.
Positivity of Anti-Neutrophil Cytoplasm Antibodies Considering the Clinical Diagnosis of the Patients for Anti-Nuclear Antibodies..
| Clinical diagnosis | Number of patients (%) | |||
|---|---|---|---|---|
| ANCA | Ethanol | Formalin cANCA | ||
| cANCA | pANCA | |||
| Systemic lupus erythematosus confirmed | 2 (9) | 1 (4) | 1 (4) | |
| Suspected ANCA associated vasculitis | 5 (50) | 1 (10) | 1 (10) | |
| Rheumatoid arthritis | 49 (63) | 2 (3) | 47 | 8 (10) |
| Autoimmune hepatitis type I | 1 (100) | 1 (100) | ||
| Total | 5700 | 4 | 50 | 8. |
ANCA: antineutrophil cytoplasmic antibodies; cANCA: cytoplasmic ANCA pattern; pANCA: perinuclear ANCA pattern.
This study found that the the probability of finding ANCA as determined by IIF assay without antigenic specificity to MPO or PR3 is greater when ANA are positive (OR=3.8, 95% CI [2.15–6.71]).
As shown in Table 2, the highest positivity for ANCA with respect to all negative ANA patients studied in each disease corresponded to patients with: UC, AAV, Sjögren's syndrome, AIH type I, RA, hepatitis, SLE and infectious disease. The presence of pANCA pattern was greater than the cANCA pattern and was observed in all diseases where these antibodies were detected except the GPA. The cANCA pattern was present in SLE, AAV, AR, infectious hepatitis, Sjögren's syndrome and UC.
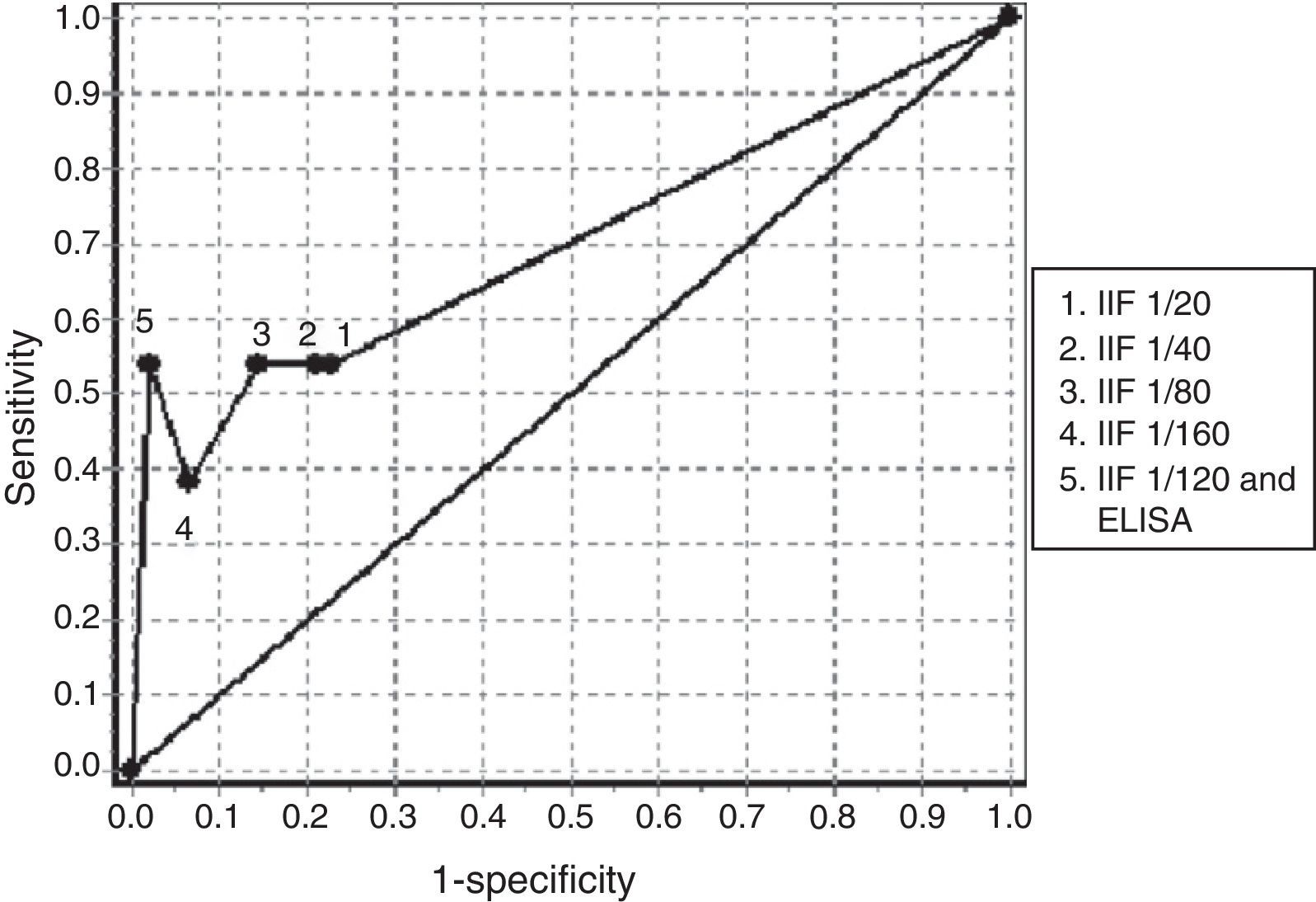
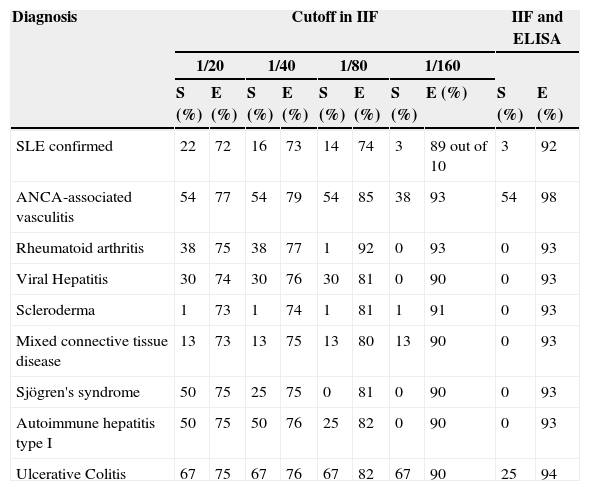
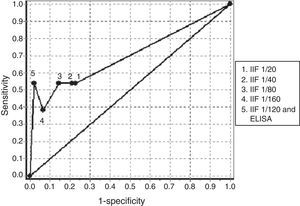
Table 4 we can see the results of sensitivity and specificity at different cutoff values obtained with ANCA determination by IIF in the group of patients with negative ANA. The AUC of the ROC curves for the determination of ANCA in the diagnosis of AAV was 0.7016 (Fig. 1). The best balance of sensitivity and specificity was obtained for patients diagnosed with AAV using the 1/80 cutoff. ANCA was determined by IIF and confirmation of anti-PR3 or anti-MPO specificity was done using ELISA according to the pattern of immunofluorescence, only showing diagnostic value for this group of diseases.
Values of Sensitivity and Specificity Obtained With Different Cutoffs in Determining Antibodies Against Neutrophil Cytoplasm in Antinuclear Antibody Negative Patients.
| Diagnosis | Cutoff in IIF | IIF and ELISA | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1/20 | 1/40 | 1/80 | 1/160 | |||||||
| S (%) | E (%) | S (%) | E (%) | S (%) | E (%) | S (%) | E (%) | S (%) | E (%) | |
| SLE confirmed | 22 | 72 | 16 | 73 | 14 | 74 | 3 | 89 out of 10 | 3 | 92 |
| ANCA-associated vasculitis | 54 | 77 | 54 | 79 | 54 | 85 | 38 | 93 | 54 | 98 |
| Rheumatoid arthritis | 38 | 75 | 38 | 77 | 1 | 92 | 0 | 93 | 0 | 93 |
| Viral Hepatitis | 30 | 74 | 30 | 76 | 30 | 81 | 0 | 90 | 0 | 93 |
| Scleroderma | 1 | 73 | 1 | 74 | 1 | 81 | 1 | 91 | 0 | 93 |
| Mixed connective tissue disease | 13 | 73 | 13 | 75 | 13 | 80 | 13 | 90 | 0 | 93 |
| Sjögren's syndrome | 50 | 75 | 25 | 75 | 0 | 81 | 0 | 90 | 0 | 93 |
| Autoimmune hepatitis type I | 50 | 75 | 50 | 76 | 25 | 82 | 0 | 90 | 0 | 93 |
| Ulcerative Colitis | 67 | 75 | 67 | 76 | 67 | 82 | 67 | 90 | 25 | 94 |
ANCA: antineutrophil cytoplasmic antibodies; E: specificity; ELISA enzyme-linked immunosorbent assay; IIF: indirect immunofluorescence; SLE: systemic lupus erythematosus; S: sensitivity.
ROC obtained with the determination of antibodies against neutrophil cytoplasm by IIF at different cutoff points and confirmation of antigenic specificities by ELISA in patients negative for antinuclear antibodies. ELISA: enzyme-linked immunosorbent assay; IIF: indirect immunofluorescence.
The values of anti-PR3 and anti-MPO antibodies were not greater than 10IU/mL in all positive cases, making them unable to compare at different cutoffs.
DiscussionMost patients included in the study, ANA negative and ANCA positive, were female. This is consistent with the increased incidence of autoimmune diseases in this gender.1
The presence of ANA was observed in a large number of patients included in the study, which coincides with the statements in the literature for this type of disease (Table 1).18 It is well known that these antibodies are present in more than 95% of patients with SLE.19
Importantly, most patients with SLE and ANCA indication were negative for ANA at a titer of 1/80, although not all patients had SLE, since a large group of patients with this diagnosis seen in this institution were not included in the study because they have no indication of ANCA. All these SLE patients considered negative for ANA presented titers below 1/80. We believe that future studies should examine the cutoff value for determination of ANA in these patients and the effective use of the diagnostic criteria for referral to an immunology laboratory.
The results obtained in patients with ANA show that the presence of ANCA can be due to the presence of antibodies against antigens different from PR3 and MPO, or interference in the ANA IIF technique. It would have been very useful in these patients to determine the presence of ANCA by ELISA against other antigens besides PR3 and MPO.
Some authors combined the IIF technique with ethanol-fixed neutrophils followed by formalin-fixed neutrophils, because the latter tends to fade when there is fluorescence positivity in ethanol produced by interference of ANA or when there are antigens different to3,6,7 MPO.
The concordance of the formalin test confirmation with respect to ELISA in the determination of anti-MPO antibodies was lower in patients with the presence of ANA. A group of these pANCA pattern patients were negative for anti-MPO antibodies and, when confirming these patterns in formalin, a cANCA pattern was obtained. This shows that the formalin test plates not always identify whether ANCA positivity in ethanol is due to the presence of antibodies against MPO.
Moreover, in a study by Pollock et al. that evaluated the use of formalin test plates to confirm the pANCA pattern in ethanol, the limitation of this technique was demonstrated in comparison with most of the MPO ELISA.6
In our opinion, faced with the presence of ANA, ANCA determination by IIF formalin plates does not provide much information.
Some authors only define the pANCA pattern on ethanol that disappears in formalin as positive for ANCA for antigens different to MPO, if the determination of ANA is negative.20
The results obtained in the Laboratory of Immunology of the NCMG show the large number of clinicopathological entities with presence of ANCA (Tables 2 and 3). The diseases that showed most ANCA positivity found in this study are largely consistent with those reported in the literature: UC (60%–80%), AIH type I (85%–95%), SLE (70%), RA without ANA (20%–30%), scleroderma, Sjögren's syndrome, polymyositis/dermatomyositis, juvenile chronic arthritis, reactive arthritis, primary sclerosing cholangitis and rapidly progressive pauci-immune glomerulonephritis (RPGN).7,9,20,21
In patients with SLE and RA, the positivity of the atypical pattern without MPO antibody specificity observed in our study is consistent with other studies where patterns are associated with atypical pANCA with antigenic specificities against lactoferrin.7,10,22
In our study a group of patients with SLE had a cANCA pattern although mostly without anti-PR3 (Table 2) antibodies. In the literature, this disease is commonly associated with cANCA. For future studies the determination of different antigenic specificities by ELISA would be very useful in these cases.
In patients with RA negative for ANA, most of the patterns obtained by IIF were pANCA without anti-MPO (Table 2) antibodies. The literature points to the presence of ANCA in RA, generally atypical pANCA, associated to antilactoferrin antibodies, although antibodies against BPE that a is associated to cANCA3,10,22 may also occur.
In patients with AIH type I negative ANA, a (Table 2) pANCA pattern was obtained. In the literature, the presence of ANCA is reported in these patients.20
In patients with CU, positivity for anti-PR3 and anti-MPO (Table 2) antibody was observed. Although studies were not found in the related literature, some authors suggest the presence of ANCA faced with PR3 due to polyclonal B cell responses during systemic inflammation associated with autoimmunity, malignancy and, in particular infections.23
In patients with Sjögren's syndrome we observed cANCA and pANCA (Table 2) patterns. The presence of ANCA has been described in these patients associated with pANCA or atypical patterns, usually with no reactivity to PR3 or against MPO. It has also been reported that antibodies can appear against cathepsin G associated to a pANCA pattern.10
The analysis of sensitivity and specificity obtained for the determination of ANCA by IIF, when a cutoff greater than 1/80 is used, is similar to that obtained in other studies in patients with AAV. The result obtained in the determination by IIF followed by confirmation by ELISA also coincides with what has been proposed in the literature (Table 4, Fig. 1).4,5
The fact that these antibodies are present in high titers in patients without AAV, when determined by IIF, but are not reactive to PR3 and MPO, confirms ELISA use in conjunction with IIF technique to establish a better diagnosis.
ANCA was shown to be present on a wide number of inflammatory and autoimmune associated states besides AAV, usually with no reactivity to PR3 and MPO. Although a pANCA pattern was observed in most patients without AAV, it is important to note the presence of a cANCA pattern, especially in patients with SLE in the population studied.
Determination of ANCA by IIF maintains its usefulness in ANA-negative patients, especially when titers at or above 1/80 are obtained, followed by determination of anti-PR3 and anti-MPO antibody by ELISA. The utility of the assay of IIF on formalin plates is less than the determination of anti-MPO antibody by ELISA when ANA are present.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflict of InterestThe authors have no disclosures to make.
Please cite this article as: Martínez Téllez G, Torres Rives B, Rangel Velázquez S, Sánchez Rodríguez V, Ramos Ríos MA, Fuentes Smith LE. Anticuerpos contra el citoplasma del neutrófilo: positividad y correlación clínica. Reumatol Clin. 2015;11:17–21.