To analyze prescription strategies followed by rheumatologist specialized in fibromyalgia (FM) treatment.
MethodsA prospective, multicentric, 3-month follow-up study with two visits. The drug treatment was started following usual clinical practice conditions. Prescription of benzodiazepines, hypnotics, anticonvulsants and antidepressants were those mainly studied. Clinical evaluation was performed by the ICAF (combined index of affection in patients with fibromyalgia) questionnaire. The most frequent strategies and their size effect were calculated.
ResultsA total of 232 patients attending 15 rheumatology centers were included. The introduction of an anticonvulsant or an antidepressant was the most common drug strategies followed in 17.7% and 14.7% of patients, respectively. Both strategies produced a statistically significant clinical improvement. The most effective strategy (with an effect size of nearly 1) was the introduction of both drugs (6.5% patients), which increased the antidepressants effect size in 50% and 100% for anticonvulsants. The strategy of not changing the number of drug prescriptions was the most frequent (35.8% patients) but no significant clinical improvement was found in these patients. Some other strategies were very uncommon and have not been analyzed in this study.
ConclusionThe introduction of anticonvulsants or antidepressants, in an isolated or combined form, produces a significant clinical improvement in FM patients. The most effective drug strategy is the introduction of both drugs at the same time. The least effective strategy is not to change the number of drug prescriptions.
Analizar las diferentes estrategias de prescripción seguidas por reumatólogos especializados en FM.
MétodosEstudio prospectivo, multicéntrico, con 2 visitas separadas 3 meses en el que se instaura un tratamiento farmacológico en condiciones de práctica clínica habitual. Se analizó fundamentalmente la prescripción de benzodiacepinas, hipnóticos, antidepresivos y anticonvulsivantes. La evaluación clínica se realizó mediante el cuestionario ICAF (Índice combinado de afectación en pacientes con fibromialgia). Se determinaron las estrategias más frecuentes y se calculó el tamaño del efecto para analizar su eficacia.
ResultadosSe incluyó a 232 pacientes con FM atendidos en 15 consultas de reumatología. Las estrategias más comunes fueron la introducción aislada de un anticonvulsivante (17,7%) o un antidepresivo (14,7), que produjeron mejorías clínicas estadísticamente significativas. La estrategia más eficaz (tamaño del efecto próximo a 1) fue la introducción simultánea de ambos fármacos (6,5%), que aumentó en un 50% el tamaño del efecto de antidepresivos y en un 100% el de anticonvulsivantes. La estrategia de no realizar ningún cambio en el número de fármacos fue la más frecuente (35,8%) pero no mostró mejoría clínica significativa. Otras estrategias fueron muy poco frecuentes y no han sido analizadas.
ConclusiónLas estrategias de prescripción de fármacos anticonvulsivantes o antidepresivos de manera aislada o en combinación producen una mejoría clínica significativa en los pacientes con FM. La estrategia más eficaz es la introducción de ambos fármacos simultáneamente. La menos eficaz es no realizar ningún cambio.
Treatment of patients with fibromyalgia (FM) is currently showing difficulties. The different treatments being used have not yet obtained the satisfactory results expected which is why patients with FM continue to maintain the same clinical manifestations even many years after being diagnosed.1
Pharmacological treatment options are quite broad because many drugs that act on the central nervous system may have beneficial effects in these patients.2,3 Analgesic, anti-inflammatory drugs (NSAIDs), antidepressants, anticonvulsants, benzodiazepines and other drugs that act on the central nervous system are used as the first line treatment in patients with FM, although in most cases are not directed towards the treatment overall disease, but only to address some of the symptoms.
However, due to the many adverse effects of these drugs also, one must be aware of what they can each contribute to the treatment of fibromyalgia and prevent overmedication, a problem often seen in these patients.
On the other hand, there are no studies to determine what type of drug is most effective, or what sequence or association should be used to obtain the best possible results.
Therefore, in this situation, when administering a drug to treat fibromyalgia not only choosing among several possibilities is the responsibility of the physician, but also knowing what strategy is the most effective.
The aim of this paper is to analyze the different strategies of drug treatment followed by rheumatologists specializing in the treatment of patients with FM and determine which ones are most effective.
MethodsPatientsMen and women over 18 years with the diagnosis of FM according to the criteria of the American College of Rheumatology4 were consecutively included, between January and April 2007. The patients came from the outpatient clinics of rheumatology of 15 different centers throughout Spain (project ICAF).
All physicians who participated in the study were selected for their special dedication to the treatment of patients with fibromyalgia. Before the study began, there was a meeting with all participating centers to standardize performance criteria between different doctors, as recommended by the consensus document of the Spanish Society of Rheumatology on fibromyalgia.2
Exclusion criteria were cardiopulmonary or other crippling diseases, morbid obesity, inflammatory rheumatic diseases and unbalanced psychiatric diseases. We also excluded those patients involved in reporting disability claims, litigation or any type of compensation processes.
The study protocol was approved by the ethics committee of clinical research at the Hospital General Universitario Gregorio Marañón in Madrid.
Study DesignThis is a prospective study with 2 separate visits over a period of 3 months for establishing a drug treatment in routine clinical practice conditions.
At the initial visit, after signing the informed consent document, the patient was interviewed face to face and sociodemographic, clinical variables were collected, as were drugs currently being used to treat fibromyalgia.
Clinical evaluation was performed using the recently validated ICAF questionnaire for our population.5,6 It consists of 4 factors: emotional, physical, active coping and passive coping as well as an overall score. ICAF assesses emotional aspects, such as anxiety and depression, and the impact they have on the patient's social environment (emotional factor). It also assesses the patient's functional capacity, fatigue, sleep quality and pain (physical factor) and the way in which the patient confronts her illness (factors of active and passive coping). As for how to score: the higher the score of each factor and the overall score, the worse the patient's clinical condition except in active coping indicating that the patient has more resources to address the disease (Annex 2 “Combined allocation index in patients with fibromyalgia (ICAF). Spanish version”).
The evaluation of drugs mainly included is that acts on the central nervous system: benzodiazepines, hypnotics, antidepressants, anticonvulsants, antihistamines, antipsychotics, and analgesics, opioids and NSAIDs.
Then the new treatment was prescribed according to usual clinical practice conditions, with the physician specialist modifying the treatment that the patient had been undergoing if they felt ill. The specialist advised all patients to undergo physical exercise in a generic way, but other non-pharmacological treatments such as psychological therapy, relaxation techniques, acupuncture, or massage diets were kept in the same preconditions that the patient had been subjected to. No other recommendation was performed and there was neither special treatment nor any limitation for prescription drug treatment in any of the centers where the study was undertaken.
At the final visit the same evaluations were performed again, same as were made at the first visit and the drugs the patient was taking again quantified.
Statistical AnalysisBefore beginning the analysis, patient data were carefully reviewed to study the distribution of frequencies and detect defects collection. Data for ICAF scores are expressed in scores T, as previously described in the work original.5
In the statistical analysis compared the number and type of drugs used in the initial visit compared to the final visit, as well as differences in the overall score and each of the factors of the ICAF.
Missing data were resolved following 2 strategies: data for the initial visit were used as vertical median for each group, and lost data from the final visit were completed by dragging the data from the initial visit, considering more conservative procedure.
We performed a descriptive statistical analysis of data on both visits and compared it using the t-test for related samples for the differences between the scores of the initial and final views.
To evaluate treatment efficacy between the two visits we calculated the effect size. The effect size is an index that expresses the magnitude of a change7; that is, an index of how much the difference between two groups is. The magnitude of the effect size is measured according to the categories proposed by Cohen, which considers a value >0.8–0.5 as large, between 0.5 and 0.2 as moderate and <0.2 as small.
We used the statistical software version 17.0 SPSS WIN for statistical analysis and considered significant a P<.05.
ResultsWe included 232 patients in the analysis, 98% women, mean age 47.7±8.6 years and duration of disease 4.3±4.0 years. All physicians provided approximately the same number of patients per study.
The number of drugs to treat FM was 3.1±1.6 drugs/patient at the initial visit, with the distribution shown in Table 1. After treatment the number of drugs rose to 3.4±1.9 drugs/patient (P<.017) in the final visit. The major changes between the two visits occurred between NSAIDs, antidepressants, benzodiazepines and anticonvulsants (Table 1).
Type of Drug Employed in 232 Patients With FM and Individual Changes Produced After Treatment in Everyday Clinical Practice Conditions.
| Initial Visit | Final Visit | Dif. % | P | |||
| Patients | % | Patients | % | |||
| Analgesics | 148 | 63.8 | 136 | 58.6 | −5.2 | ns |
| NSAID | 132 | 56.9 | 105 | 45.3 | −11.6 | .0001 |
| Antidepressants | 118 | 50.9 | 167 | 72.0 | +21.1 | .0001 |
| Benzodiazepines | 104 | 44.8 | 75 | 32.3 | −12.5 | .0001 |
| Anticonvulsants | 34 | 14.7 | 96 | 41.4 | +26.7 | .0001 |
| Hypnotics | 14 | 6.0 | 20 | 8.6 | +2.6 | ns |
| Other | 8 | 3.4 | 13 | 5.6 | +2.2 | ns |
ns=non significant.
For further analysis we only took into account drugs that act directly on the central nervous system and analgesics and NSAIDs were discarded. Moreover, the consumption of hypnotics has added the use of benzodiazepines, since in many cases they are also used to induce sleep.
The most significant changes were: an increase in the number of antidepressants, from 0.50±0.50 to 0.71±0.45 (P<.0001) antidepressants/patient an increase number of anticonvulsants, from 0.14±0.35 to 0.41±0.49 (P<.0001) anticonvulsants/patient, and a reduction in the number of benzodiazepines, from 0.48±0.50 to 0.38±0.48 (P<.001) benzodiazepines/patient. Regarding the number of drugs introduced into treatment only one was introduced in 87 patients in 15 patients 2 were entered and in 4 patients 3 drugs were introduced simultaneously.
From the point of view of clinical manifestations, there was a significant improvement between the initial and the final visits affecting the emotional and physical factors, active coping and overall score in the total group of patients.
Specific Strategies for Change in Treatment and its ConsequencesSpecific strategies for change in the treatment followed by physicians in the study were to add an anticonvulsant alone (17.7% patients), adding an antidepressant alone (14.7% patients) or add both drugs simultaneously (6.5% of patients).
However, it is noteworthy that the most common strategy was to stop all treatment in 35.8% of patients. Within this group, 26% of patients not taking medication for central nervous system, but 25% of them took one, 36% took 2 and 11% took 3 of the drugs.
The remaining strategies were varied and were used in 25.3% of patients. In many cases, the strategy involved the suspension of a drug replaced by the introduction of another, so it was difficult to assess their effectiveness individually.
At the initial visit, we found no statistically significant differences between treatment groups in the baseline variables studied: age, sex, duration of disease, time since diagnosis or employment status. There were also no differences in baseline total score of ICAF or any of its four factors (Table 2).
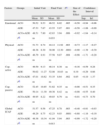
Differences in Factors and Global ICAF Scores Between Groups of Patients in Which an Anticonvulsant (ACO; n=41), an Antidepressant (ADE; n=34) or Both Drugs Simultaneously Were Introduced (ACO+ADE; n=15). The Table Shows Values at the Initial Visit, Final Visit and the Size of the Effect Between Both.
| Factors | Groups | Initial Visit | Final Visit | Pa | Size of the Effect | Confidence Interval | |||
| Mean | SD | Mean | SD | Sup. | Inf. | ||||
| Emotional | ACO | 50.52 | 9.53 | 46.52 | 6.02 | .005 | −0.50 | −0.94 | −0.06 |
| ADE | 47.53 | 7.67 | 43.55 | 5.97 | .001 | −0.58 | −1.06 | −0.09 | |
| ACO+ADE | 48.51 | 7.80 | 42.83 | 3.94 | .006 | −0.92 | −1.64 | −0.14 | |
| Pb | ns | 0.033 | |||||||
| Physical | ACO | 51.73 | 8.74 | 44.12 | 11.92 | .003 | −0.73 | −1.17 | −0.27 |
| ADE | 48.36 | 8.30 | 38.90 | 12.30 | .0001 | −0.90 | −1.39 | −0.39 | |
| ACO+ADE | 46.79 | 8.91 | 37.56 | 9.87 | .008 | −0.98 | −1.71 | −0.20 | |
| Pb | ns | ns | |||||||
| Cop. active | ACO | 49.50 | 9.13 | 48.14 | 8.38 | ns | −0.16 | −0.59 | 0.28 |
| ADE | 50.02 | 11.27 | 52.08 | 10.83 | ns | 0.19 | −0.29 | 0.66 | |
| ACO+ADE | 47.01 | 10.62 | 53.35 | 8.64 | .002 | 0.65 | −0.10 | 1.37 | |
| Pb | ns | ns | |||||||
| Cop. passive | ACO | 52.45 | 10.85 | 51.62 | 9.35 | ns | −0.08 | −0.51 | 0.35 |
| ADE | 50.14 | 11.30 | 49.36 | 8.42 | ns | −0.08 | −0.55 | 0.40 | |
| ACO+ADE | 49.95 | 8.22 | 49.85 | 6.79 | ns | −0.01 | −0.73 | 0.70 | |
| Pb | ns | ns | |||||||
| Global ICAF | ACO | 51.57 | 9.56 | 47.25 | 8.78 | .003 | −0.48 | −0.91 | −0.03 |
| ADE | 48.29 | 8.75 | 42.23 | 9.03 | .0001 | −0.68 | −1.16 | −0.19 | |
| ACO+ADE | 49.36 | 10.34 | 41.04 | 5.94 | .003 | −0.99 | −1.72 | −0.20 | |
| Pb | ns | 0.013 | |||||||
ACO: anticonvulsant; ADE: antidepressant; Cop.: coping; SD: standard deviation; ns: non significant.
When considering the therapeutic measures taken subsequently, we analyzed two situations: the presence or absence of changes. When comparing patients in which a drug was discontinued (n=26) with those where a drug was introduced (n=102), the only significant difference was that the emotional factor scored higher (worse emotional score) compared to the group of patients where a drug was introduced (49.77±8.87 vs 54.45±9.92, P=.021), but no significant differences in other factors or the overall score.
The group of patients who underwent the strategy and in whom the treatment was not modified, there was no additional benefits for patients. Overall ICAF score and its factors were not significantly changed in this group of patients between the initial and final visits.
In the group of patients in whom the strategy was to add only an anticonvulsant, there were significant differences in emotional and physical factors as well as the overall score with a moderate effect size (Table 2).
At the other group of patients in which only an antidepressant was added, there were also significant differences in the overall score and the emotional and physical factors with a slightly higher effect size than in the previous group (Table 2). Note that the greatest effect among the factors was achieved in the physical component of ICAF.
Finally, in the patient group in which the strategy was the introduction of both drugs, antidepressant and anticonvulsant effects were the greatest of all figures with effect size close to 1 (Table 2).
While the strategy of adding an antidepressant or an anticonvulsant was generally made exclusively, a change in strategy with benzodiazepines was not as smooth. These were added and suspended exclusively in about the same number of patients, but also changes in benzodiazepines more often accompanied the introduction or withdrawal of the other 2 drugs. Regardless of changes in the other 2 drugs, overall benzodiazepines were discontinued in 36 patients and were added as new drug in only 3 patients. The analysis of the strategy to discontinue benzodiazepines produced an improvement in the overall score and in all factors except passive coping (data not shown).
DiscussionIn this paper we have analyzed some of the most common strategies of drug administration followed by a group of physicians with special attention to the treatment of patients with FM, in conditions of medical practice. The results suggest that certain changes in the drug delivery strategy are accompanied by significant changes in clinical status of patients.
As a general strategy, there was a significant increase in the total number of drugs used to treat patients and more often involve the introduction of a single drug.
Some of the treatment strategies were used more often than others by the physicians of this study, and among them, strategies to individually incorporate an antidepressant, an anticonvulsant or both drugs simultaneously were clearly more frequent. Other strategies, for example stopping benzodiazepines, were used, and although there were indications that it may also be beneficial, it could not be tested due to the small number of patients in this situation since this strategy was accompanied, often, in exchange for some other drugs.
However, the strategy of not making changes to the treatment was the most frequent of all affecting 36% of patients. However, in these patients the clinical evaluation of the initial and final visits showed no significant differences in the clinical situation of patients.
There remains considerable skepticism regarding FM8 and a fairly widespread opinion among physicians is that drug treatment does not provide much benefit so the patients are often only treated with drugs to treat symptoms of pain, mainly analgesics and NSAIDs.
One possible explanation may lie in the fact that in the European Union there is no indication of drug treatment in FM, while in the U.S., the FDA3 has approved 2 drugs, dual antidepressants and inhibitors of serotonin and norepinephrine (duloxetine and milnacipram), and an anticonvulsant drug (pregabalin) for this indication. Therefore, in the European Union, these drugs can only be given when dealing with clinical manifestations for which they are approved; however, in many FM patients these clinical manifestations are not exactly the most relevant.
In this sense, the finding in our study that the only difference between patients to whom drugs were introduced and those to which the drug was discontinued, emotional factors, which measure anxiety and depression, confirm that these drugs were prescribed to treat these symptoms and not others such as pain, fatigue, and functional capacity as measured by the physical factor.
A meta-analysis has shown that various recent9 antidepressants produce a particularly favorable effect on pain, sleep, mood, fatigue and mood status changes, but with a low effect size in relation to perceived quality of life. Something similar happens with anticonvulsants, which have also shown that there is a degree of hard evidence that pregabalin and gabapentin produced improvement in pain and sleep, but not on fatigue or mood alterations.10
In our work we found that administration of an antidepressant or an anticonvulsant individually improves the patient's general condition with a moderate effect size. However, when added both at the same time the effect size increases by almost 50% compared to antidepressants and up to 100% with respect to anticonvulsants. This finding is consistent with that recently suggested by some authors.11
However, we cannot rule that the mere change of drug treatment, adding 2 new drugs, could have a positive impact beyond their specific effect. The change itself and the expectations of the patient before can have a therapeutic effect.12
It is noteworthy that the major effect of these drugs occurs precisely on the physical factor of ICAF followed the emotional factor. The factors of active and passive coping did not change significantly, as there are ways in which patients cope with the disease and are not modified in the short term by the introduction of a drug.6
The criterion to follow a particular strategy has not been entirely uniform among physicians in the study. Thus, the strategy to not modify the treatment was followed by physicians in 36% of patients despite their clinical situation in terms of degree of impairment was similar to that of other patients who used other strategies. However, within this group of patients, 74% were receiving one or more drugs so the scope for any type of change was large.
Moreover, there was a similar number of patients to whom only benzodiazepines were suspended or added, suggesting that some strategies are not shared unanimously by all physicians.
The main limitation of this study is that it has not been able to evaluate all possible strategies of drug administration but only those most frequently followed by doctors, and we cannot conclude on which is the most effective strategy. Originally the study was not designed for it so the results are only for guidelines on clinical practice in the sample of clinical units involved in the study.
Another limitation is that some variables were not analyzed in this study and yet may behave as potential confounders. Variables such as comorbidity, or treatment followed for this, and were not evaluated in our study and may affect results obtained in relation to the effectiveness of different strategies.
Although it initially was not recommended that researchers change the preconditions of psychological therapy or physical exercise, it is possible that over the 3-month follow-up study period there was some sort of modification that may have influenced the results.
We note that the importance of this study is that it allows us to know which are the main strategies of prescription drugs for fibromyalgia patients followed in our environment and in conditions of medical practice. Although data from our study cannot ensure the best possible strategy, we found some evidence to suggest that among the most common strategies some of them might be superior to others. These findings should be tested in future studies.
FinancingThis study was financed by the Fondo de Investigación Sanitaria (FIS)PI 07/0202, of the Health and Consumption ministry.
Conflict of InterestsPfizer Pharmaceuticals contributed economically to the development of the ICAF project as a research Project of the Spanish Foundation for Rheumatology. The 3 authors of the study have been principal investigators of the ICAF project.
ICAF group: Alegre C. (Hospital Vall de Hebrón, Barcelona), Belenguer R. (Hospital 9 de Octubre, Valencia), Belmonte M. (Hospital General de Castellón, Castellón), Beltrán J. (Hospital General de Castellón, Castellón), Blanch J. (Hospital IMAS, Barcelona), Collado A. (Hospital Clínic, Barcelona), Fernández Dapica P. (Hospital Universitario 12 de Octubre, Madrid), Francisco Hernández F.M. (Hospital Dr. Negrín, Gran Canaria), García Monforte A. (Hospital Universitario Gregorio Marañón, Madrid), González Hernández T. (IPR, Madrid), González Polo J. (Hospital Universitario La Paz, Madrid), Hidalgo C. (Centro Reumatológico, Salamanca), Mundo J. (Hospital Clínic, Barcelona), Muñoz Carreño P. (Hospital General, Guadalajara), Vallejo I. (Hospital Clínic, Barcelona), Vidal J. (Hospital General, Guadalajara).