We report one case of dermatomyositis and one of polymyositis refractory to several conventional inmunosupressive therapies, which present a response after treatment with rituximab, enabling steroid dose reduction and a prolonged remission.
Se presentan un caso de dermatomiositis y otro de polimiositis refractarias a varios inmunosupresores convencionales y con respuesta a tratamiento con rituximab, con el que se posibilita la disminución de dosis de corticoide y se mantiene a la enfermedad en remisión durante un largo periodo.
Idiopathic inflammatory myopathies (IIM) are connective tissue diseases of unknown etiology which primarily affect skeletal muscle, skin and other internal organs. Glucocorticoids (GC) and immunosuppressants such as methotrexate (MTX) and azathioprine (AZA) have been used for treatment with a high percentage of inneficacy.1 Different biologic therapies, including rituximab (RTX), a chimeric anti CD20 monoclonal antibody, have occasionally been used. We report a case of refractory dermatomyositis (DM) and polymyositis (PM) intolerant to multiple conventional treatments that later responded to RTX.
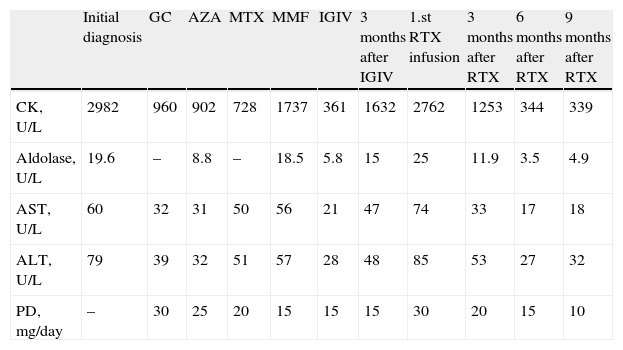
Clinical CasesCase 1The patient is a 50 year old woman with a history of smoking and depression, diagnosed with DM in 2008 with the presence of a skin rash on the face and arms, proximal limb muscle weakness, elevated muscle enzymes (creatine kinase [CK]: 2982 U/aldolase l: 19.6U/L) and a positive ANA with a titer of 1/640 and negative specific autoantibodies. The electromyogram shows a proximal myopathy of widespread distribution and moderate intensity. She had gastric intolerance and hypertension associated with high doses of GC, and had initially been prescribed prednisone (PD) at 0.5mg/kg/day, with a decrease in the CK values (960U/L) but without improvement of muscle weakness. Subsequent muscle enzymes and the dose of PD with respect to the patient receiving immunosuppressive therapy are shown in Table 1. AZA 150mg/day and MTX 15mg/week were suspended due to gastric intolerance with neither producing significant clinical improvement. Mycophenolate mofetil (MMF) 2g/day was suspended due to inefficiency and worsening clinical and laboratory. After use, the patient was reluctant to undergo an unconventional and unapproved treatment for MII. Because of the appearance of Cushing's syndrome features and cataracts, along with persistent disease activity, in 2010 it was decided to treat the patient with intravenous immunoglobulin (IVIG) at doses of 2g/kg/month for 3 months. There was a good clinical and analytical response but with a rapid relapse after 3 months of suspension. Thus, in February 2011 she was treated with RTX (2 iv infusion cycles of 1g separated by 2 weeks) without other immunosuppressives, showing improvement in muscle weakness and a near normalization of muscle enzymes. This response occured 5 months after the first infusion, but was maintained over time after 9 months without need for retreatment and facilitated a reduction in PD to 10mg/day. During this monitoring no adverse effects related to RTX were detected.
Case 1: Progression of Muscles Enzymes and Treatment.
| Initial diagnosis | GC | AZA | MTX | MMF | IGIV | 3 months after IGIV | 1.st RTX infusion | 3 months after RTX | 6 months after RTX | 9 months after RTX | |
| CK, U/L | 2982 | 960 | 902 | 728 | 1737 | 361 | 1632 | 2762 | 1253 | 344 | 339 |
| Aldolase, U/L | 19.6 | – | 8.8 | – | 18.5 | 5.8 | 15 | 25 | 11.9 | 3.5 | 4.9 |
| AST, U/L | 60 | 32 | 31 | 50 | 56 | 21 | 47 | 74 | 33 | 17 | 18 |
| ALT, U/L | 79 | 39 | 32 | 51 | 57 | 28 | 48 | 85 | 53 | 27 | 32 |
| PD, mg/day | – | 30 | 25 | 20 | 15 | 15 | 15 | 30 | 20 | 15 | 10 |
GC: glucocorticoids; AZA: azathioprine; MTX: methotrexate; MMF: mycophenolate mofetil; IGIV: intravenous immunoglobulin; RTX: rituximab; CK: creatinkynase (normal: <250); aldolase (normal: <7); AST: aspartate aminotransferase (normal: 10–40); ALT: alanine aminotransferase (normal: 10–40); PD: prednisone.
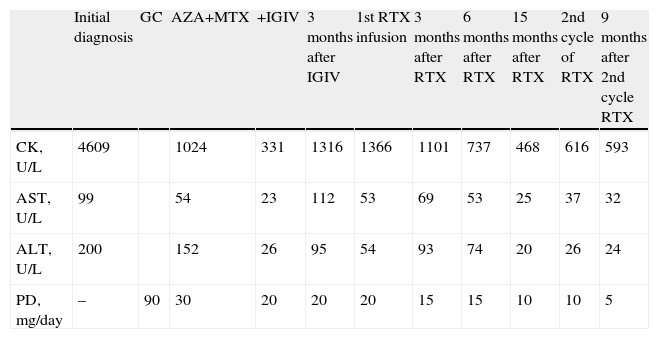
This patient is a 30-year-old woman with a history of hypertension and morbid obesity, evaluated in 2007 due to proximal limb weakness and elevated muscle enzymes (CK: 4609U/L). PM was diagnosed by the demonstration in the electromyogram of a diffuse proximal myopathy on the muscle biopsy and pathological data consistent with inflammatory myopathy. Initially, PD 1mg/kg/day was employed with a partial response with a decreasing dose to 0.5mg/kg/day after addition of immunosuppressants. These did not prevent several episodes of generalized weakness and severe functional disability requiring treatment with boluses of methylprednisolone (1g/day for 3 consecutive days). The evolution of the values of muscle enzymes and treatment is reflected in Table 2. Although MTX (20mg/day) and AZA (150mg/day) was employed, it was impossible to decrease the dose of PD under 30mg/day because of clinical deterioration, so IGIV (2g/kg/month for 3 months) was started, producing clinical and laboratory improvement, but relapsed after 3 months of its termination despite maintaining AZA and MTX associated with GC. In August 2009 it was decided to use RTX (two iv infusions of 1g separated by 2 weeks), maintaining GC, AZA and MTX. At 5 months, there is a great improvement in the proximal weakness and with a decrease in muscle enzymes, which was maintained over time. In January 2011, and after 17 months of the first cycle of RTX, she was given a second course for fear of a relapse, because the patient had to travel abroad for a long time and it was impossible to follow up. With this new dose, she remained in remission for a year and is still in remission, with an almost normalization of CK values (468U/L) and a dose of PD of 5mg/day. No side effects were related to the infusion of RTX or during follow up.
Case 2: Progression of Muscle Enzymes and Treatment.
| Initial diagnosis | GC | AZA+MTX | +IGIV | 3 months after IGIV | 1st RTX infusion | 3 months after RTX | 6 months after RTX | 15 months after RTX | 2nd cycle of RTX | 9 months after 2nd cycle RTX | |
| CK, U/L | 4609 | 1024 | 331 | 1316 | 1366 | 1101 | 737 | 468 | 616 | 593 | |
| AST, U/L | 99 | 54 | 23 | 112 | 53 | 69 | 53 | 25 | 37 | 32 | |
| ALT, U/L | 200 | 152 | 26 | 95 | 54 | 93 | 74 | 20 | 26 | 24 | |
| PD, mg/day | – | 90 | 30 | 20 | 20 | 20 | 15 | 15 | 10 | 10 | 5 |
GC: glucocorticoids; AZA: azathioprine; MTX: methotrexate; IGIV: intravenous immunoglobulin; RTX: rituximab; CK: creatinkynase (normal: <250); AST: aspartate aminotransferase (normal: 10–40); ALT: alanine aminotransferase (normal: 10–40); PD: prednisone.
The IIM are characterized by the presence of a mild inflammatory infiltrate of the muscle where B lymphocytes seem to have an important role in the pathogenesis, especially in DM. RTX is a monoclonal anti-CD20 antibody used to treat non-Hodgkin lymphoma and rheumatoid arthritis, but is also used in other refractory autoimmune diseases such as SLE, Sjögren's syndrome or vasculitis, producing a profound and sustained depletion of CD20+B cells and without affecting stem cells or plasma cells. Currently, treatment of PM/DM is largely empirical and there is no agreement on what the most optimal therapy is. Initially high-dose GC is used and in cases of resistance to these, or serious extramuscular manifestations, immunosuppressants such as AZA, MTX, cyclosporine A, tacrolimus, MMF or cyclophosphamide2 are added, with around 25% of patients not responding or relapsing even immunosuppresants.3 IVIG has been tested (dose of 1–2g/kg) and shown to act quickly and improve muscle weakness, with few adverse effects but with a high cost and a transitory4 effect. Drugs that inhibit TNF-alpha do not appear to be effective, so are not recommended for use in these diseases, unless other treatment options have failed.5 There are isolated cases or small series published demonstrating the efficacy of RTX in the treatment of refractory IIMs.6–9 In these publications, a high percentage of patients who respond adequately or do not need a new cycle of early retreatment are those who received IVIG or cyclophosphamide in combination with RTX. There are also differences in the dose of RTX used, with no significant influence on the results (375mg/weekly for 4 doses or cycles of 1g every two weeks for 2 doses), and concomitant therapy used in conjunction with RTX. Garcia et al.10 collected data from 17 patients with IIM which used RTX plus cyclophosphamide IV and where there was an initial improvement in all of them (including 4 in whom respiratory disease was present), requiring a second course of RTX at 11 months only in 5 patients, with subsequent favorable responses in all cases. The publication with the largest number of IIM cases treated with RTX refractory was published by Couderc et al.,11 based in the French AIR (Auto-Immunity and Rituximab) trial. They obtained a favorable response in 16 of the 30 patients with refractory IIM treated with RTX, with an average efficiency of 15 months and a decrease in the values of CPK and dose of GC in most of them. The dose used was 1g×2 doses 2 weeks apart in 83% of cases and 375mg/m2 weekly×4 weeks in 17%. Most received concurrent GC and immunosuppressive treatment (28 and 21 patients, respectively) and of 10 patients a favorable response was seen in 8 of them.
We expose the effectiveness of RTX in refractory DM and PM (to immunosuppresants) but without the simultaneous use of IVIG or cyclophosphamide and allowing lower doses of GC without relapse. We used a dose of RTX similar to that employed for rheumatoid arthritis by checking their effectiveness in other publications. In both cases, the response to RTX was late (5 months or so) but, unlike the response induced by IVIG, remained constant for at least 1 year after the first cycle.
Regarding the possible side effects of RTX, there are several, the most frequent being infusional reactions and other hematological disorders as well as infections (upper respiratory, urinary or skin) or severe and progressive multifocal leukoencephalopathy. In our 2 cases, administration of RTX was well tolerated and not accompanied by adverse effects.
ConclusionBased on the cases presented and previously published experiences, we believe that RTX is a valid alternative to consider in the treatment of DM and PM refractory to conventional treatment, even without other simultaneous immunosuppressive drugs. The dose of RTX and the optimal treatment for remission should be established.
Ethical disclosuresProtection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this investigation.
Confidentiality of Data. The authors declare that they have followed the protocols of their work centre on the publication of patient data and that all the patients included in the study have received sufficient information and have given their informed consent in writing to participate in that study.
Right to privacy and informed consent. The authors have obtained the informed consent of the patients and /or subjects mentioned in the article. The author for correspondence is in possession of this document.
DisclosuresThe authors have no disclosures to make.
Please cite this article as: Sánchez-Fernández SÁ, et al. Eficacia de rituximab en dermatomiositis y polimiositis refractarias al tratamiento convencional. Reumatol Clin. 2012. http://dx.doi.org/10.1016/j.reuma.2012.02.007.