Osteogenesis imperfecta (OI) is an inherited connective tissue disease. The disease has been linked to mutations in one of the type I collagen genes. The diagnosis is based on clinical and radiologic findings. The management of OI in adults is not well-established and includes physical rehabilitation, surgical procedures, the use of antiresorptive therapy and anabolic agents. The aim of the present work was to analyze the clinical and analytical characteristics of these patients in adulthood, as well as to evaluate the different treatments administered. We reviewed the cases of OI diagnosed in our centre over the last 12 years (2005–2017). We describe 15 adult patients with OI.
La osteogénesis imperfecta (OI) es un trastorno hereditario del tejido conectivo generalmente relacionado con mutaciones de los genes del colágeno tipo I. El diagnóstico se basa en los hallazgos clínicos y radiológicos. El manejo clínico de la OI en adultos no está del todo establecido y comprende desde la rehabilitación física y los procedimientos quirúrgicos hasta el uso de tratamientos antirresortivos y osteoformadores. El objetivo del presente trabajo ha sido analizar las características clínicas y analíticas de estos pacientes en la edad adulta, así como evaluar los diferentes tratamientos administrados. Se han revisado los casos de OI diagnosticados en nuestro centro en los últimos 12 años (2005-2017). Se describen 15 pacientes adultos con OI.
Due to its low rate of incidence(1/10,000–1/25,000 newborns), osteogenesis imperfecta (OI), belongs to the group of rare diseases. It has traditionally been considered an autosomal dominant (AD) bone dysplasia caused by mutations in type I collagen genes but the discovery of new causative genes, mainly recessive, supports a physiopathology predominantly related to collagen disorders.1,2
Type I collagen is a structural component of the extracellular matrix of the connective tissue whose function is to provide support and resistance to tissues. The majority of OI cases (90%) come from heterozygous mutations in one of the genes encoding pro-collagen I (COL1A1, collagen type, α-1 and COL1A2, collagen type I, α-2)1,2 peptic chains.
In 1979 Sillence and Rimoin suggested an OI classification based on clinical, radiographic and genetic findings. This classification distinguishes between: OI type I (mild with blue sclera), OI type II (perinatal lethal form), OI type III (severe and progressively deforming with normal sclera) and OI type IV (moderate severity with normal sclera).3 At present, the finding of new genes involved and phenotypical heterogenicity in the mutations of the collagen I gene has led to the appearance of up to 17 subtypes.4 Although there is no universally accepted classification due to the high genetic complexity and to extreme phenotypical variability, the nosology group of the International Skeletal Dysplasias Society recommends maintaining the Sillence classification to stage the level of OI severity.5
OI diagnosis is based on clinical and radiologic findings. The main characteristics are fractures from mild traumas, accompanied by deformities in the long bones and in the chest, scoliosis, kyphosis and delayed growth. Since this is a general connective tissue disorder, we may find different extra-skeletal symptoms, such as blue-tinged whites of the eyes, dentinogenesis imperfecta, loss of hearing, impairment of lung capacity and alterations in heart valves.1
Bone densimetry is a widely validated test in postmenopausal osteoporosis but few studies analyze the measurement of bone mass in adults with OI.6,7 Although bone mineral density (BMD) may be an indicator of the severity of the disease according to some studies,8 at present its main use is monitoring changes to bone mass after treatment.
In OI bone histomorphomology reveals a reduction of bone volume and of the number of trabeculae, with a raised bone turnover.1,2
OI management is multidisciplinary and mainly involves physical and orthopaedic surgery therapy.2 With regard to pharmacological therapy, although there are several efficacious drugs available for the prevention of fractures in postmenopausal women and in male osteoporosis, such as biphosphonates, the anti-RANKL monoclonal antibody (denosumab) and the parathyroid PTH 1–34 (teriparatide)6,9 hormone in OI the defect in the bone matrix does not necessarily respond to these pharmacological agents and its effect on the reduction of fractures is yet to be demonstrated.10 In children with moderate to severe OI with a history of multiple long bone fractures and/or vertebral fractures the use of biphosphonates is broadly extended even in early ages.2,11 However, treatment in adults with moderate to severe OI is much less established because there are few studies with low patient numbers available.1,12,13 Although it has been demonstrated in paediatrics biphosphonates increase lumbar BMD and improve vertebral height and geometry,10,14,15 there is limited evidence of fracture prevention. A recent Cochrane review which included 8 random studies with a total of 403 patients concluded that treatment with oral or intravenous biphosphonates increased BMD. Four studies show a drop in fractures in patients treated with biphosphonates. However, no significant differences were observed in 3 studies included and in the remaining study a drop in vertebral and in upper extremity fractures was observed, but not in the lower extremities.10
In osteoporosis, teriparatide (PTH) is associated with an anabolic bone response, with an increase in formation and BMD, an improvement in microarchitecture and a reduction in fracture risk.16 Some observational studies describe a positive effect of teriparatide on bone mass in patients with type I OI.17 Recently, Orwoll et al. carried out a randomized trial compared with placebo and observed a significant increase of BMD in patients who received teriparatide, without demonstrating differences in the presence of fractures.18
Other therapies used in OI which are undergoing research are denosumab (anti-RANKL monclonal antibody), gene therapy or stem cell transplant therapy.1,6,9,19,20
Bone fractures and deformity mainly occur in infancy, but the risk of fracture remains high into adult life, when the deleterious effect of age overlaps with bone fragility of the disease. This fact demonstrates the importance of regular follow-up check-ups with these patients.
The aim of this review was to analyze the clinical and analytical features of our patients with OI in adults and assess the different treatments administered.
Patients and methodA retrospective study was conducted of patients diagnosed with OI referred from the Rheumatology Service of the University Hospital Dr. Peset for the last 12 years (from 2005 up until 2017). A total of 15 patients with OI were included. In all cases clinical symptoms were reported; age when diagnosed; laboratory data; radiologic report; treatment and disease evolution.
With regard to the laboratory study, the phosphocalcic metabolism was considered, including the levels of calcium, phosphorous, alkaline phosphatase, parathyroid hormone and levels of 25-hydroxycholecalciferol (25OHD) and bone remodelling markers. The bone formation marker analyzed was P1NP (procollagen type I terminal of telopeptide) and the bone resorption marker was β-CTX (collagen type I C-terminal telopetide). In cases when it was possible BMD (g/cm2) was analyzed at lumbar spine level L2–L4 and at total femur level. Osteoporosis was diagnosed in compliance with World Health Orgnaisation (WHO).
ResultsFifteen patients with OI (4 men and 11 women) were included in the study aged between 22 and 60 years, with a mean age of 44 years. In the majority of patients (n=11) the disease began in childhood with repeated fractures and bone deformities. All patients had a history of fragility fractures, in 6 cases hip fracture and in 4 bilateral. Eight patients had family histories of OI. Of the 6 patients who underwent the genetic study (massive and Sanger sequencing of gene COL1A2 and gene COL1A2), only 2 had mutations in the collagen genes, one case in COL1A1 and the other in COL1A2. According to the Sillence classification, 7 patients presented with OI type I, 3 patients OI type III, 2 patients OI type iv and 3 patients were not classified.
The 4 patients who were diagnosed with OI as adults were referred from the Trauma Department for study after presenting with a fracture. The other patients had been diagnosed in childhood and were referred from the Rheumatology department on reaching adulthood for follow-up, from the Endocrinology and Paediatric Services.
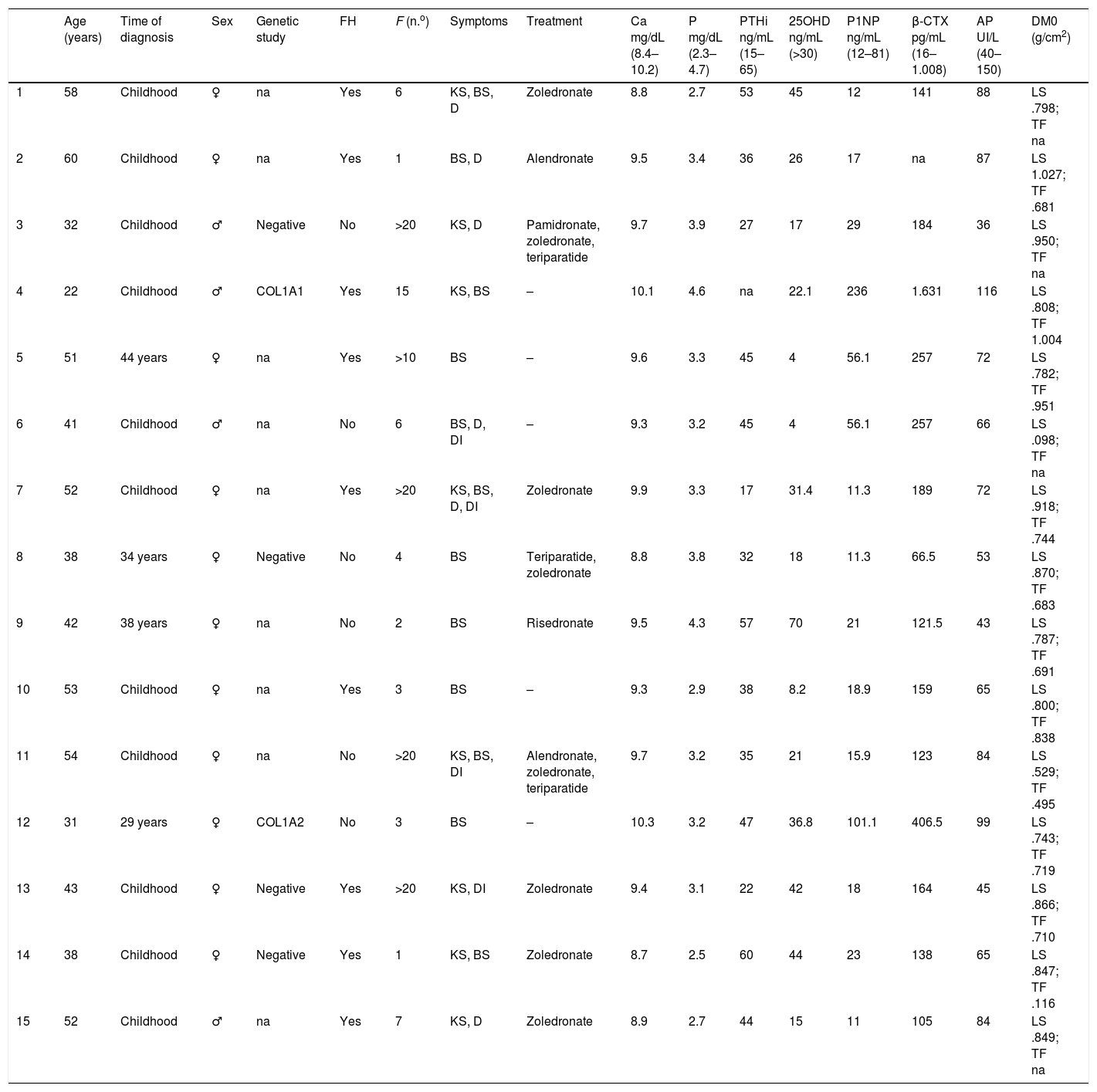
Clinical features and laboratory dataAfter the fractures, the most common medical symptom was the presence of blue sclera (11), followed by kyphoscoliosis (8), deafness (6) and dentinogenesis imperfecta (3). Eight patients required help with crutches or wheelchairs, 3 and 5 cases respectively. Data are contained in Table 1.
Patient characteristics and laboratory data.
| Age (years) | Time of diagnosis | Sex | Genetic study | FH | F (n.o) | Symptoms | Treatment | Ca mg/dL (8.4–10.2) | P mg/dL (2.3–4.7) | PTHi ng/mL (15–65) | 25OHD ng/mL (>30) | P1NP ng/mL (12–81) | β-CTX pg/mL (16–1.008) | AP UI/L (40–150) | DM0 (g/cm2) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 58 | Childhood | ♀ | na | Yes | 6 | KS, BS, D | Zoledronate | 8.8 | 2.7 | 53 | 45 | 12 | 141 | 88 | LS .798; TF na |
| 2 | 60 | Childhood | ♀ | na | Yes | 1 | BS, D | Alendronate | 9.5 | 3.4 | 36 | 26 | 17 | na | 87 | LS 1.027; TF .681 |
| 3 | 32 | Childhood | ♂ | Negative | No | >20 | KS, D | Pamidronate, zoledronate, teriparatide | 9.7 | 3.9 | 27 | 17 | 29 | 184 | 36 | LS .950; TF na |
| 4 | 22 | Childhood | ♂ | COL1A1 | Yes | 15 | KS, BS | – | 10.1 | 4.6 | na | 22.1 | 236 | 1.631 | 116 | LS .808; TF 1.004 |
| 5 | 51 | 44 years | ♀ | na | Yes | >10 | BS | – | 9.6 | 3.3 | 45 | 4 | 56.1 | 257 | 72 | LS .782; TF .951 |
| 6 | 41 | Childhood | ♂ | na | No | 6 | BS, D, DI | – | 9.3 | 3.2 | 45 | 4 | 56.1 | 257 | 66 | LS .098; TF na |
| 7 | 52 | Childhood | ♀ | na | Yes | >20 | KS, BS, D, DI | Zoledronate | 9.9 | 3.3 | 17 | 31.4 | 11.3 | 189 | 72 | LS .918; TF .744 |
| 8 | 38 | 34 years | ♀ | Negative | No | 4 | BS | Teriparatide, zoledronate | 8.8 | 3.8 | 32 | 18 | 11.3 | 66.5 | 53 | LS .870; TF .683 |
| 9 | 42 | 38 years | ♀ | na | No | 2 | BS | Risedronate | 9.5 | 4.3 | 57 | 70 | 21 | 121.5 | 43 | LS .787; TF .691 |
| 10 | 53 | Childhood | ♀ | na | Yes | 3 | BS | – | 9.3 | 2.9 | 38 | 8.2 | 18.9 | 159 | 65 | LS .800; TF .838 |
| 11 | 54 | Childhood | ♀ | na | No | >20 | KS, BS, DI | Alendronate, zoledronate, teriparatide | 9.7 | 3.2 | 35 | 21 | 15.9 | 123 | 84 | LS .529; TF .495 |
| 12 | 31 | 29 years | ♀ | COL1A2 | No | 3 | BS | – | 10.3 | 3.2 | 47 | 36.8 | 101.1 | 406.5 | 99 | LS .743; TF .719 |
| 13 | 43 | Childhood | ♀ | Negative | Yes | >20 | KS, DI | Zoledronate | 9.4 | 3.1 | 22 | 42 | 18 | 164 | 45 | LS .866; TF .710 |
| 14 | 38 | Childhood | ♀ | Negative | Yes | 1 | KS, BS | Zoledronate | 8.7 | 2.5 | 60 | 44 | 23 | 138 | 65 | LS .847; TF .116 |
| 15 | 52 | Childhood | ♂ | na | Yes | 7 | KS, D | Zoledronate | 8.9 | 2.7 | 44 | 15 | 11 | 105 | 84 | LS .849; TF na |
♀: male; ♂: female; 25OHD: 25-hydroxycholecalciferol; FH: family history; β-CTX: C-terminal of telopeptide of type I collagen; Ca: calcium; KS: kyphoscoliosis; LS: lumbar spine L1–L4; COL1A1: collagen type I, alph-1; COL1A2: collagen type I, alpha-2; DI: dentinogenesis imperfecta; BMD: bone mineral density; BS: blue sclera; F (n.o): fracture (number); AP: alkaline phosphatase; TF: total femur; D: deafness; na: not available; P: phosphorous; P1NP: terminal of telopeptide of type I collagen; PTHi: parathormone intact.
All the patients diagnosed in childhood presented with fractures during this period and 7 of them also presented with new fractures as adults (3 received biphosphonates). The 4 patients diagnosed as adults have had fractures.
According to densitometric criteria, all patients presented with low bone mass; 11 with osteoporosis criteria and 4 with osteopenia. In 4 patients it was not possible to carry out exploration of the hip.
Levels of calcium, phosphorous and alkaline phosphatase were normal in all patients. With regard to the bone remodelling markers, P1NP was normal in 5 patients, low in 4 and high in the other 3; the β-CTX was normal in the majority (n=13) and high in 2 patients; levels of parathyroid intact hormone (PTHi) were normal in almost all patients (n=12). Most patients (n=9) presented with insufficient levels of 25-hydroxycholecalciferol (<30ng/ml).
TreatmentFrom time of diagnosis all patients had initiated physiotherapy treatment, physical treatment and some had required orthopaedic surgery. However, only 2 patients had received pharmacological treatment during childhood with intravenous zoledronic acid or pamidronic acid.
Current treatment of patients is based on the use of biphosphonates. Six patients received treatment with intravenous zoledronic acid and 2 with oral biphosphonates (alendronic and risedronic acid). Another 2 patients had started treatment with subcutaneous teriparatide after presenting with fractures despite the biphosphonate. At present 5 patients are not receiving any specific treatment.
DiscussionOI is a rare genetic disease which is usually diagnosed in childhood, but it must be taken into consideration when there is a presence of fractures in young patients without any other secondary causes of osteoporosis.1,2
In a period of 12 years 15 patients were assessed with a diagnosis of OI with bone deformities and pathological fractures as the most common form of presentation.
There are few studies that assess the role of the BMD in the diagnosis or prediction of fracture risk in adult patients with OI. Although the densitrometry is not necessary for establishing the diagnosis of OI, it may be a marker of disease severity and may be a predictor of functional capacity long term.8 Many studies describe the difficulty in analysing the results of mineral density due to the considerable deformities presented by the patients. Adults with OI often present with scoliosis, which makes it difficult to assess the spine features. Furthermore, there is no consensus on how to define osteoporosis in adults with OI. In our series using the WHO criteria we found there was a low bone mass in all patients and in the majority in osteoporosis (73%).
In the patients with OI calcium levels are generally normal and vitamin D levels may be low, especially for those who are in wheelchairs.7,21 Also, a rise in bone remodelling has been described. In our series the patients generally presented with normal levels of calcium and of bone remodelling markers, but vitamin D deficiency was notably high.
Although there are no available studies which analyze optimal duration of therapy with biphosphonates, their long term side effects and their role in the prevention of fractures they are currently used as the main pharmacological treatment of OI.1,6,9 All patients treated have received biphosphonates but if they presented with new fractures despite this treatment, teripariatide treatment was initiated. Studies with teriparatide are providing promising results for patients with OI, although it appears that the response would vary depending on the type of OI.1,18 Some published data with the anti-RANKL monoclonal antibody (denosumab) may herald a new useful therapy for these patients.2,6,9,19
OI is a multifarious disease which requires a multidisciplinary approach. It appears that treatment with biphosphonates and teriparatide increases bone mass but further studies are required to assess their effect on fractures.
Conflict of interestsThe authors have no conflict of interests to declare.
Please cite this article as: Díaz López M, Alegre Sancho JJ, Martínez-Ferrer À. Osteogénesis imperfecta. Descripción de 15 casos. Reumatol Clin. 2020;16:165–168.