To describe the history, clinical features and microorganisms involved in a group of adult subjects with and without septic arthritis (SA) at a tertiary care in Mexico.
Material and methodsA cross-sectional descriptive study was conducted on 96 adults with clinical suspicion of AS in one or more joints. In all cases synovial fluid arthrocentesis and culture was performed. The comparison group subjects were culture negative. A descriptive statistical analysis and binary logistic regression model was performed between the variables associated with the development of AS. A value of P≤.05 was significant.
ResultsA total of 49 out of 96 subjects had a positive culture, mostly of the monoarticular type (96%; P=.02). The knee was the most common site (61%; P=.06) and pain was the main clinical manifestation (59%; P=.001). Staphylococcus was the most common etiological agent (65%; P<.001). The risk factors revealed in the final regression model were SA the history of joint disease (OR=25; P=.03) and volume increase (OR=13.16; P=.06). Functional limitation (OR=8.54; P=.04) showed a significant risk among borderline for SA.
ConclusionsOur results are consistent with previous studies, and can be generalized to geographical areas with similar clinical features to those observed in this study.
Describir los antecedentes, características clínicas y microorganismos implicados en un grupo de sujetos adultos con y sin artritis séptica (AS) en un centro de tercer nivel de atención en México.
Material y métodosDiseño descriptivo, de tipo transversal analítico en 96 adultos con sospecha clínica de AS en una o más articulaciones. En todos los casos se realizó artrocentesis y cultivo de líquido sinovial. El grupo de comparación fueron sujetos con cultivo negativo. Un análisis con estadística descriptiva y un modelo con regresión logística binaria se realizó entre las variables asociadas al desarrollo de AS. Un valor de p≤0,05 fue significativo.
ResultadosUn total de 49/96 sujetos tuvieron cultivo positivo, la mayoría del tipo monoarticular (96%; p=0,02), principalmente la rodilla (61%; p=0,06). Las manifestaciones clínicas incluyeron: aumento de volumen (57%; p=0,01), dolor (59%; p=0,001) y limitación de la función (51%; p=0,002). El estafilococo fue el agente etiológico más común (65%; p<0,001). El modelo de regresión final reveló como factores de riesgo para AS el antecedente de enfermedad articular (RM=2,33), osteoartritis (RM=25,04) y uso de corticoides (RM=5,25). Clínicamente, el aumento de volumen (RM=13,16), el dolor (RM=5,79) y la limitación funcional (RM=8,54) se mostraron con un riesgo entre limítrofe y significativo para AS.
ConclusionesNuestros resultados son congruentes con estudios previos y pueden generalizarse a zonas geográficas con características clínicas similares a las observadas en este estudio.
Septic arthritis is a joint disease that can affect one or more joints in patients of any age.1 It is considered a medical emergency2 and, despite the new diagnostic techniques, the morbidity has changed little over the last 25 years.3 In Europe, the incidence is estimated to range between 2 and 6 cases per 100000 population4; this number can be multiplied by 10 among individuals with osteoarthritis or rheumatoid arthritis,5 those with a joint replacement, older persons, alcoholics, diabetics, immunocompromised patients, individuals with skin ulcers, those treated with intraarticular corticosteroids or synthetic materials6 and in indigenous populations.7
The organisms that cause septic arthritis differ from one part of the world to another.8 In the majority of the countries of Europe and North America, a greater frequency of staphylococci and streptococci has been reported.9 Gram-negative bacteria may be associated with septic arthritis in up to 20% of the cases, mainly when immunosuppression, cancer, diabetes or cirrhosis is involved.10 The diagnosis of septic arthritis is suspected on the basis of the clinical picture (joint pain, functional limitation and body temperature >38.5°C) and is confirmed by means of the gold standard, that is, a positive joint fluid culture.
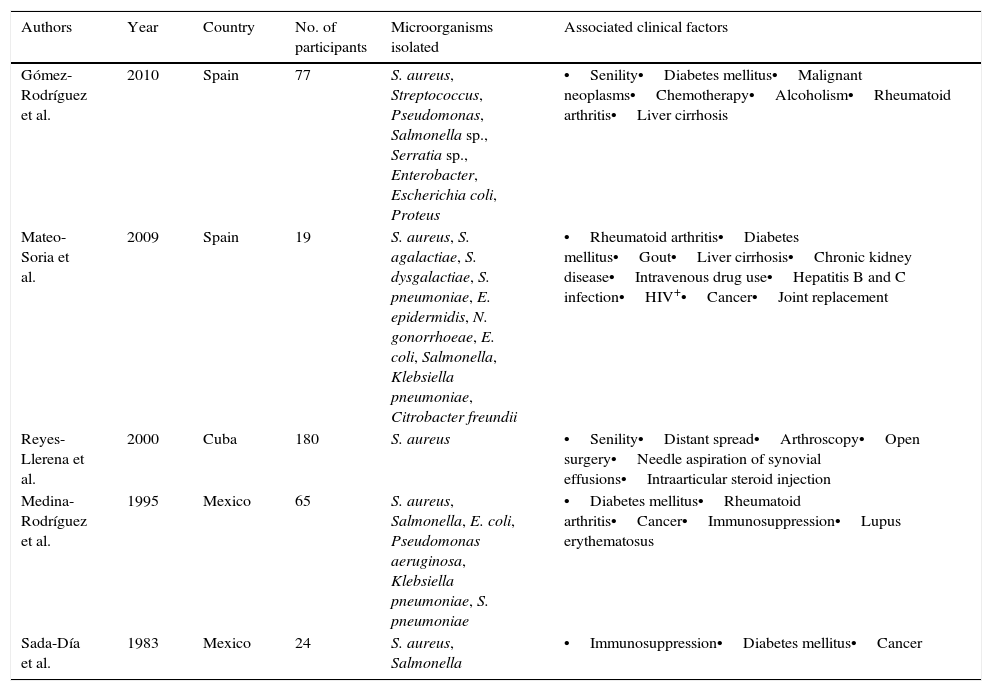
In Mexico and the rest of Latin America, few studies have focused on septic arthritis from the perspective of comparing the major known risk factors to enable the application of this knowledge to similar geographic regions. Table 1 compares the microorganisms and clinical factors that have been associated with them in some of these studies. For the purpose of describing the medical history, clinical characteristics and microorganisms involved in a group of adults with and without a diagnosis of septic arthritis, we carried out a prospective comparative study in a tertiary care center in Mexico.
Comparison of Microorganisms Isolated and Associated Clinical Factors in Studies of Septic Arthritis in Mexico, Cuba and Spain.
| Authors | Year | Country | No. of participants | Microorganisms isolated | Associated clinical factors |
|---|---|---|---|---|---|
| Gómez-Rodríguez et al. | 2010 | Spain | 77 | S. aureus, Streptococcus, Pseudomonas, Salmonella sp., Serratia sp., Enterobacter, Escherichia coli, Proteus | •Senility•Diabetes mellitus•Malignant neoplasms•Chemotherapy•Alcoholism•Rheumatoid arthritis•Liver cirrhosis |
| Mateo-Soria et al. | 2009 | Spain | 19 | S. aureus, S. agalactiae, S. dysgalactiae, S. pneumoniae, E. epidermidis, N. gonorrhoeae, E. coli, Salmonella, Klebsiella pneumoniae, Citrobacter freundii | •Rheumatoid arthritis•Diabetes mellitus•Gout•Liver cirrhosis•Chronic kidney disease•Intravenous drug use•Hepatitis B and C infection•HIV+•Cancer•Joint replacement |
| Reyes-Llerena et al. | 2000 | Cuba | 180 | S. aureus | •Senility•Distant spread•Arthroscopy•Open surgery•Needle aspiration of synovial effusions•Intraarticular steroid injection |
| Medina-Rodríguez et al. | 1995 | Mexico | 65 | S. aureus, Salmonella, E. coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, S. pneumoniae | •Diabetes mellitus•Rheumatoid arthritis•Cancer•Immunosuppression•Lupus erythematosus |
| Sada-Día et al. | 1983 | Mexico | 24 | S. aureus, Salmonella | •Immunosuppression•Diabetes mellitus•Cancer |
HIV, human immunodeficiency virus.
A cross-sectional, descriptive analysis was conducted to study 96 adults of both sexes with suspected septic arthritis, recruited among patients seeking care in a hospital-based orthopedic outpatient clinic, who presented with some clinical sign of pain, inflammation, reddening, increased warmth or reduced mobility in one or more joints. The study was carried out in a tertiary care center in northeastern Mexico between January 2010 and December 2012. Authorization was granted by the research ethics committee of the hospital prior to the initiation of the study. All the patients provided their written informed consent before enrolling in the study.
Participant RecruitmentArthrocentesis and synovial fluid culture were performed in every case. Patients whose synovial fluid was considered to be contaminated or whose clinical data were incomplete were excluded, as were those who did not choose to participate in the study. The septic arthritis group consisted of 49 individuals whose clinical picture included at least two of the following symptoms in one or more joints: increased sensitivity, pain, erythema or functional limitation, and who were carriers of a bacterial agent isolated in culture. The comparison group was made up of 47 individuals with a similar clinical picture and with a leukocyte count in synovial fluid lower than 50000/mm3, as well as absence of microorganisms in the synovial fluid.
Measurements and Case ReviewsWe designed a tool that comprised more than 40 variables which we obtained from the medical records and personal interviews with our patients. They included demographic data relative to hospital admission and discharge, known risk factors for septic arthritis (injuries, comorbidities and use of corticosteroids, among others), possible origin of the infection and clinical symptoms. During the clinical evaluation, synovial fluid was collected from the affected joint, and was analyzed by means of Gram stain and bacterial culture. The causative agent was defined as that which was reported in the results of the culture and was classified as gram-positive or gram-negative. Finally, the clinical and laboratory data were compared and included in the final analysis.
Declaration of Ethical PrincipalsThe study was approved by the hospital research ethics committee and we followed the research protocols established by the institution in which the study was carried out with regard to gaining access to the clinical data of the patients. All of the information was obtained for the only purpose of its analysis and subsequent publication in scientific media.
Statistical AnalysisThe categorical variables were expressed in terms of frequency distribution and percentages, and measures of central tendency (mean) and variability (standard deviation) were used for the continuous variables. The categorical variables were compared by means of Pearson's chi-square (χ2) and Fisher's test, when the expected frequencies were less than 5. Odds ratio (OR), estimated from 2×2 contingency tables, was utilized to identify those variables associated with septic arthritis. The variables that proved to have a P value <.200 were included in a binary logistic regression model. Of all the variables analyzed, those with the highest P value were excluded from the model, and logistic regression analysis was again carried out using the remaining variables. This process was repeated successively, and was completed when all the variables utilized in the model proved to be significant (P<.05). The results were expressed as OR with 95% confidence intervals (CI). The consistency of the model was evaluated on the basis of the area under the receiver operating characteristic (ROC) curve, and the assumptions of the model were tested by means of the calculation of residues. The statistical analysis was performed using version 19 of the SPSS® statistical software package for Windows (SPSS, Chicago, IL, United States). A P value less than or equal to .05 was considered to indicate statistical significance.
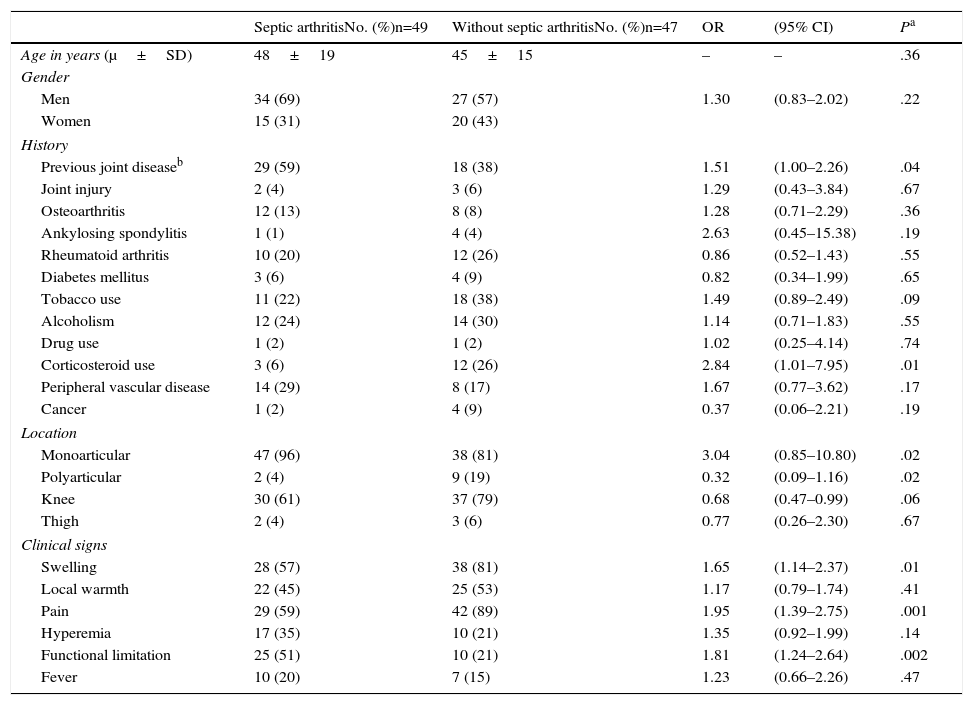
ResultsSociodemographic and Clinical Characteristics of the SampleThe study group consisted of 96 individuals with a mean age of 46.83±16.68 years (P=.36). The diagnosis of septic arthritis was confirmed in 49 patients (51%), whose mean age was 48±19 years. In contrast, those individuals without septic arthritis were relatively younger.
The incidence of septic arthritis was higher among the men (69%), in contrast to the women, in whom the prevalence observed was significantly lower (31%). A history of joint disease (injury, previous surgery) was recorded in 59% of the patients with septic arthritis and was associated with significant risk (OR=1.51; 95% CI, 1.00–2.26; P=.04). It is important to point out that there was clear evidence of a history of some joint injury that could explain the development of infection in only two cases (OR=1.29; 95% CI, 0.43–3.84]; P=.67). In all, 39% of the patients had a history of some type of rheumatic disease: osteoarthritis, ankylosing spondylitis or rheumatoid arthritis. We observed no significant association between any of these conditions and a higher risk for septic arthritis.
The use of corticosteroids was positively associated with the risk of developing septic arthritis (OR=2.84; 95% CI, 1.01–7.95; P=.01). In contrast, no significant association was observed for a history of diabetes mellitus, tobacco use, alcohol consumption, intravenous drug use, peripheral vascular disease or the presence of cancer.
With regard to the localization of the septic arthritis, in most cases, a single joint was involved (96%; P=.02), there being an evident higher risk in these patients (OR=3.04; 95% CI, 0.85–10.80). Knees were the most frequently affected joints (61%; P=.06).
The clinical signs associated with septic arthritis included: swelling (57%; OR=1.65; 95% CI, 1.14–2.37; P=.01], pain (59%; OR=1.95; 95% CI, 1.39–2.75; P=.001] and functional limitation (51%; OR=1.81; 95% CI, 1.24–2.64]; P=.002). Other manifestations that indicated a diagnosis of septic arthritis but were not statistically significant were: hyperemia (35%; OR=1.35; 95% CI, 0.92–1.99; P=.14), increased local warmth (45%; OR=1.17; 95% CI, 0.79–1.74; P=.41) and fever (20%; OR=1.23; 95% CI, 0.66–2.26; P=.47). The general characteristics and age ranges of the study participants with and without septic arthritis are shown in Table 2.
General Characteristics of the Individuals Studied With and Without Septic Arthritis.
| Septic arthritisNo. (%)n=49 | Without septic arthritisNo. (%)n=47 | OR | (95% CI) | Pa | |
|---|---|---|---|---|---|
| Age in years (μ±SD) | 48±19 | 45±15 | – | – | .36 |
| Gender | |||||
| Men | 34 (69) | 27 (57) | 1.30 | (0.83–2.02) | .22 |
| Women | 15 (31) | 20 (43) | |||
| History | |||||
| Previous joint diseaseb | 29 (59) | 18 (38) | 1.51 | (1.00–2.26) | .04 |
| Joint injury | 2 (4) | 3 (6) | 1.29 | (0.43–3.84) | .67 |
| Osteoarthritis | 12 (13) | 8 (8) | 1.28 | (0.71–2.29) | .36 |
| Ankylosing spondylitis | 1 (1) | 4 (4) | 2.63 | (0.45–15.38) | .19 |
| Rheumatoid arthritis | 10 (20) | 12 (26) | 0.86 | (0.52–1.43) | .55 |
| Diabetes mellitus | 3 (6) | 4 (9) | 0.82 | (0.34–1.99) | .65 |
| Tobacco use | 11 (22) | 18 (38) | 1.49 | (0.89–2.49) | .09 |
| Alcoholism | 12 (24) | 14 (30) | 1.14 | (0.71–1.83) | .55 |
| Drug use | 1 (2) | 1 (2) | 1.02 | (0.25–4.14) | .74 |
| Corticosteroid use | 3 (6) | 12 (26) | 2.84 | (1.01–7.95) | .01 |
| Peripheral vascular disease | 14 (29) | 8 (17) | 1.67 | (0.77–3.62) | .17 |
| Cancer | 1 (2) | 4 (9) | 0.37 | (0.06–2.21) | .19 |
| Location | |||||
| Monoarticular | 47 (96) | 38 (81) | 3.04 | (0.85–10.80) | .02 |
| Polyarticular | 2 (4) | 9 (19) | 0.32 | (0.09–1.16) | .02 |
| Knee | 30 (61) | 37 (79) | 0.68 | (0.47–0.99) | .06 |
| Thigh | 2 (4) | 3 (6) | 0.77 | (0.26–2.30) | .67 |
| Clinical signs | |||||
| Swelling | 28 (57) | 38 (81) | 1.65 | (1.14–2.37) | .01 |
| Local warmth | 22 (45) | 25 (53) | 1.17 | (0.79–1.74) | .41 |
| Pain | 29 (59) | 42 (89) | 1.95 | (1.39–2.75) | .001 |
| Hyperemia | 17 (35) | 10 (21) | 1.35 | (0.92–1.99) | .14 |
| Functional limitation | 25 (51) | 10 (21) | 1.81 | (1.24–2.64) | .002 |
| Fever | 10 (20) | 7 (15) | 1.23 | (0.66–2.26) | .47 |
%, percentage; CI, confidence interval; No., number of patients; OR, odds ratio; SD, standard deviation.
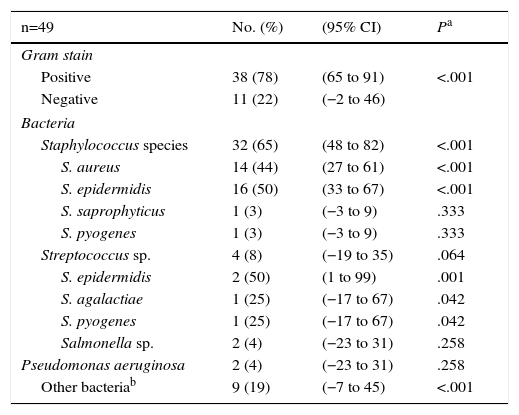
Gram staining was performed in 49 cases; of these, 78% were gram-positive (95% CI, 65–91; P<.001). The study of the synovial fluid identified staphylococcus as the most frequently isolated microorganism (65% of the cases; 95% CI, 48–82; P<.001). Among the cocci isolated in culture, those most frequently found were Staphylococcus aureus (S. aureus) (44%; 95% CI, 27–61; P<.001) and Staphylococcus epidermidis (50%; 95% CI, 33–67; P<.001). Gram-negative bacteria were identified in few cases (22%). Among those isolated were Salmonella sp. (4%; 95% CI, −23 to 31; P=.258) and Pseudomonas aeruginosa (4%; 95% CI, −23 to 31; P=.258). Other bacteria isolated either alone or in combination with those mentioned above included: Stenotrophomonas maltophilia, Enterococcus faecalis, Escherichia coli, Klebsiella pneumoniae, Actinomyces sp. and Chryseobacterium indologenes. None of our patients had septic arthritis due to Neisseria gonorrhoeae. The results of the bacteriological analysis of the joint fluid collected from the patients with septic arthritis are shown in Table 3.
Bacteriologic Analysis of Joint Fluid of Patients With Septic Arthritis.
| n=49 | No. (%) | (95% CI) | Pa |
|---|---|---|---|
| Gram stain | |||
| Positive | 38 (78) | (65 to 91) | <.001 |
| Negative | 11 (22) | (−2 to 46) | |
| Bacteria | |||
| Staphylococcus species | 32 (65) | (48 to 82) | <.001 |
| S. aureus | 14 (44) | (27 to 61) | <.001 |
| S. epidermidis | 16 (50) | (33 to 67) | <.001 |
| S. saprophyticus | 1 (3) | (−3 to 9) | .333 |
| S. pyogenes | 1 (3) | (−3 to 9) | .333 |
| Streptococcus sp. | 4 (8) | (−19 to 35) | .064 |
| S. epidermidis | 2 (50) | (1 to 99) | .001 |
| S. agalactiae | 1 (25) | (−17 to 67) | .042 |
| S. pyogenes | 1 (25) | (−17 to 67) | .042 |
| Salmonella sp. | 2 (4) | (−23 to 31) | .258 |
| Pseudomonas aeruginosa | 2 (4) | (−23 to 31) | .258 |
| Other bacteriab | 9 (19) | (−7 to 45) | <.001 |
%, percentage; CI, confidence interval; No., number of patients; OR, odds ratio.
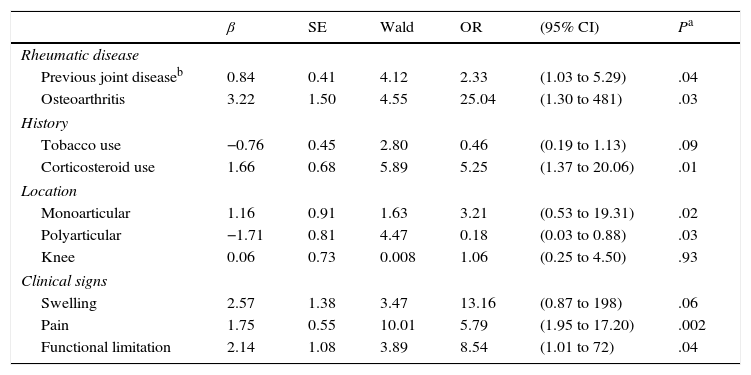
Binary logistic regression analysis was performed in those variables in which the initial univariate analysis demonstrated some significant association with the diagnosis of septic arthritis. Thus, from the statistical point of view, the final model identified the presence of previous joint disease (OR=2.33; 95% CI, 1.03–5.29; P=.04) and osteoarthritis (OR=25.04; 95% CI, 1.30–481; P=.03) as risk factors for the development of septic arthritis. Among the background features described, the use of corticosteroids persisted as a risk factor for the development of septic arthritis (OR=5.25; 95% CI, 1.37–20.06; P=.01).
Likewise, the involvement in the disease of a single joint (OR=3.21; 95% CI, 0.53–19.31; P=.02) was associated with the diagnosis of septic arthritis. Finally, with respect to the clinical signs, the presence of pain (OR=5.79; 95% CI, 1.95–17.20; P=.002) and functional limitation (OR=8.54; 95% CI, 1.01–72; P=.04) were shown to be predictors associated with the diagnosis of septic arthritis. In contrast, swelling (OR=13.16; 95% CI, 0.87–198; P=.06) was associated with borderline risk. The results of the binary logistic regression analysis in relation to the clinical signs, comorbidities and location of the septic arthritis can be found in Table 4.
Results of the Adjusted Analysis of the Characteristics Observed in Patients With and Without Septic Arthritis Using Binary Logistic Regression.
| β | SE | Wald | OR | (95% CI) | Pa | |
|---|---|---|---|---|---|---|
| Rheumatic disease | ||||||
| Previous joint diseaseb | 0.84 | 0.41 | 4.12 | 2.33 | (1.03 to 5.29) | .04 |
| Osteoarthritis | 3.22 | 1.50 | 4.55 | 25.04 | (1.30 to 481) | .03 |
| History | ||||||
| Tobacco use | −0.76 | 0.45 | 2.80 | 0.46 | (0.19 to 1.13) | .09 |
| Corticosteroid use | 1.66 | 0.68 | 5.89 | 5.25 | (1.37 to 20.06) | .01 |
| Location | ||||||
| Monoarticular | 1.16 | 0.91 | 1.63 | 3.21 | (0.53 to 19.31) | .02 |
| Polyarticular | −1.71 | 0.81 | 4.47 | 0.18 | (0.03 to 0.88) | .03 |
| Knee | 0.06 | 0.73 | 0.008 | 1.06 | (0.25 to 4.50) | .93 |
| Clinical signs | ||||||
| Swelling | 2.57 | 1.38 | 3.47 | 13.16 | (0.87 to 198) | .06 |
| Pain | 1.75 | 0.55 | 10.01 | 5.79 | (1.95 to 17.20) | .002 |
| Functional limitation | 2.14 | 1.08 | 3.89 | 8.54 | (1.01 to 72) | .04 |
β, standardized coefficient; CI, confidence interval; OR, odds ratio; SE, standard error.
For the purpose of describing the clinical characteristics, microorganisms involved and comorbidities in a group of adults with septic arthritis, we reviewed the cases of acute arthritis in a tertiary care center in northeastern Mexico. A total of 96 patients in whom septic arthritis was suspected were studied. Ultimately, 49 cases were confirmed and were included as cases that were positive for septic arthritis, and the remainder, as a comparison group.
At the present time, it is increasingly common to find cases of polyarticular septic arthritis associated with rheumatoid arthritis, chronic diseases, antimicrobial resistance, aging or immunosuppression.3,11 However, similar to the findings reported by other authors,12 the majority of the cases in this study involved a single joint, with the knee occupying first place. A number of hypotheses have been proposed to explain this fact; however, the one most widely accepted is associated with hematogenous spread or the proximity of bacteria that penetrate the articular fluid because of the absence of a basement membrane in the synovial tissue following a knee injury or lesion.13
Among the clinical and epidemiological factors associated with the development of bacterial septic arthritis, we mentioned age, as up to 40% of the cases occur in adults over the age of 60 years.14 Among the risk factors that increase the probability of diagnosing an acute joint inflammation such as septic arthritis, those that are always included are age, history of diabetes mellitus, rheumatoid arthritis, recent joint surgery, joint replacement (synthetic material) and skin infection, among others.15
Although gender has been reported to be a factor of higher risk for certain specific types of septic arthritis, this aspect cannot be generalized. The majority of the individuals with septic arthritis in this study were men, a finding similar to those reported in previous studies.16
Another important factor in the development of septic arthritis is a history of an underlying rheumatic disease (lupus, osteoarthritis, rheumatoid arthritis or gout), principally in cases of polyarticular arthritis.17 In our study group, 13% of the cases were associated with osteoarthritis and 20% with rheumatoid arthritis. Binary logistic regression analysis revealed a statistically significant association for the presence of osteoarthritis. It has been pointed out that osteoarthritis, together with injuries,18 are a frequent cause of septic arthritis in patients over the age of 50 years,19 and more so if they are associated with the presence of autoimmune diseases,20 drug use and corticosteroids.18
Among the background features, the use of corticosteroids has been shown to be an important risk factor for the development of septic arthritis. We should not overlook the fact that the use of corticosteroids was established in previous studies and our patients were no exception.21
The etiology and clinical characteristics of septic arthritis are not easy to study, especially because it is difficult to establish an accurate bacteriological diagnosis in a large number of cases.22 The culture of the joint fluid is the gold standard for the diagnosis of infectious arthritis, with a sensitivity that ranges from 75% to 97%.23–25 In this respect, the microbiological pattern of septic arthritis has not changed much, and in recent decades, the majority of the researchers continue to highlight S. aureus and Streptococcus as the major causative agents,3,16 mainly because bacterial colonization is favored when there is tissue damage, which exposes the synovial membrane to contamination.26,27
In most hospitals, the evaluation of an inflamed joint can be carried out in a simple manner, with the examination of the synovial fluid, looking for crystals and bacteria, using a Gram stain, which has a sensitivity of nearly 60%.28 In this study, 78% of the participants had a positive Gram stain which pointed to the diagnosis and made it possible to start antibiotic therapy in a timely fashion. This is the policy in the center in which we work, committed to the Mexican Guidelines for the Prevention, Diagnosis and Treatment of Acute Septic Arthritis in Children and Adults (IMSS-368-10).29
As reported by other authors, staphylococci and streptococci were the infectious agents most frequently isolated.30 There was little presence of gram-negative bacteria, as agents of that type were detected in only four cases. There was a notable absence of cases of gonococcal arthritis, in contrast to the situation reported in North America and Australia, where it is a common cause of septic arthritis in young adults.22,23 In contrast, in European countries, the incidence is very low, similar to the findings in our series.11 As a possible explanation for this circumstance, Hook et al. proposed the existence of variations in the capacity of Neisseria gonorrhoeae to cause joint damage in different geographical regions of the world, a hypothesis that has not yet been confirmed by any study.16,22,31 Recently, Oppegaard et al.32 have reported the high specificity (98%) of CD64 and procalcitonin for the diagnosis of infectious diseases, including septic arthritis. As yet we have no experience in this regard in our setting and the possibility of including these diagnostic tests among others that could be utilized for the analysis of septic arthritis remains open.
A detailed clinical history and well performed physical examination are sufficient to awaken the suspicion of septic arthritis within the first 3 days of onset of symptoms in the majority of cases.33 In this respect, swelling, the presence of pain and functional limitation were the most prevalent clinical signs in our population and, in the logistic regression model, were clinical manifestations that enabled us to reach the diagnosis. Eder et al.8 and, somewhat later, Margaretten et al.10 reported pain to be a consistent symptom in up to 85% of the patients with septic arthritis. However, the individual differences in pain threshold can influence the final diagnosis. This was made clearer in a study performed by Burgos-Vargas et al. in 1210 patients with septic arthritis in which the objective was to predict the diagnosis of septic arthritis using a multivariate analysis that included pain (scored according to a visual analog scale), functional disability and the use of nonsteroidal anti-inflammatory drugs. At the end of the study, the diagnosis was based on a pain score of 5.2 or higher out of 10 possible points (maximum pain)34; thus, the authors conclude that, during the clinical evaluation of a painful, inflamed joint, a thorough analysis of all the signs in the inflamed joint would point toward the diagnosis and make it possible to rule out other possible causes of nonseptic arthritis.
Functional limitation continues to be a relevant clinical sign in septic arthritis. Our study found a prevalence of 51%; previous studies report values that range from 10% to 73%, in which the patient characteristics, type of bacterium, a correct and timely diagnosis and the treatment received had a direct influence on its development.16 Logistic regression analysis revealed a significant increase in the risk for swelling and functional limitation as important factors in the diagnosis of septic arthritis. This information agrees with the idea that the finding of a swollen, inflamed, reddened and overly warm joint is reason enough to include septic arthritis in the differential diagnosis of a joint disease.3 In our results, we observed no significant differences for the increase in the local temperature or hyperthermia, a fact that could be more due to the samples size of our study population than to a lack of statistical association.
In the case of fever, Margaretten et al. report that it has a low sensitivity (57%) for the diagnosis of septic arthritis10; consequently, it is considered to be a symptom of low specificity for the majority of conditions involving joints.35–37
Finally, synovial biopsy is known to be of no use in the diagnosis of acute bacterial arthritis; however, when an unclear diagnosis is suspected, it is common for the physician to perform a biopsy in cases of chronicity of the inflammatory signs, and in agreement with the clinical history of the patient in cases of amyloidosis, sarcoidosis or suspected metastatic tumors.38 In our center, it is performed only in patients who do not respond to medical treatment, when there is some doubt concerning the diagnosis or chronicity of the clinical picture, as we mentioned above.
This study has a number of limitations: it was carried out in a single center, the sample size, the cross-sectional (but not prospective) design, the fact that it was not possible to perform a cytological analysis of the synovial fluid in every case, an analysis was not done with additional tests for other diseases associated with septic arthritis in the group of individuals with negative bacterial culture, it was not possible to carry out a follow-up of the treatment and of the complications because many patients were lost to follow-up in our center when they returned to the municipalities from which they had been referred. However, the exclusion of synovial fluids considered to be contaminated and the methodology utilized allow us to suppose that the number of cases studied is representative of the region and that the findings can be applied to other areas or hospitals in which the population is similar. Moreover, it lends support to the results of previous studies in terms of the risk factors and clinical characteristics of septic arthritis.
In conclusion, as has been reported by other researchers, the most common clinical presentation of septic arthritis continues to be monoarthritis of the knee, associated with gram-positive bacteria, in which evidence of an inflamed, reddened and overly warm joint is reason enough to consider this diagnosis. Additional prospective studies will be required to further evaluate the course, treatment and complications of septic arthritis in Mexican patients.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
The author wishes to thank the Mexican Social Security Institute for permitting our access to the information in clinical records.
Please cite this article as: Ornelas-Aguirre JM. Artritis séptica en un centro de adultos de tercer nivel de atención. Reumatol Clin. 2016;12:27–33.