Lung ultrasound (LUS) is a clinical and research tool with great potential in the diagnosis and monitoring of diffuse interstitial lung disease (ILD) present in systemic autoimmune diseases (SAD). Appropriate training in LUS is essential for the correct and safe use of this technique.
ObjectiveTo document the current state of LUS education and use among Spanish rheumatologists and pneumologists.
Material and MethodsA national online survey was designed for members of the Spanish Society of Rheumatology and the ILD Area of the Spanish Society of Pneumology and Thoracic Surgery. The survey consisted of 22 questions on demographics, professional activity, performance and training in LUS.
ResultsOne hundred and thirty-five (56.72% rheumatologists, 41.79% pneumologists) responded to the survey. Of these, 56.30% were part of an ILD Unit in their centre. LUS in clinical practice was performed by 35.82% but only 14.93% performed it in ILD, mainly for diagnostic purposes. Training in LUS of responders had been diverse in format, content and sponsors. The vast majority (87.79%) considered that the optimal model of education in LUS should be standardized and structured and consist of a combination of theoretical-practical courses and the conduct of a minimum number of supervised LUS examinations, with competency assessment.
ConclusionsThe current lack of formal structured education in LUS is an opportunity to develop quality educational programmes in this emerging field.
La ecografía pulmonar (ECOPULM) es una herramienta clínica y de investigación con un gran potencial en el diagnóstico y seguimiento de las enfermedades pulmonares intersticiales difusas (EPID) presentes en las enfermedades autoinmunes sistémicas (EAS). La formación apropiada en ECOPULM es esencial para el uso correcto y seguro de esta técnica.
ObjetivoConocer la formación y uso de la ECOPULM entre los reumatólogos y neumólogos españoles.
Material y métodosSe diseñó una encuesta nacional en línea, dirigida a los socios de la Sociedad Española de Reumatología y del Área de EPID de la Sociedad Española de Neumología y Cirugía Torácica. La encuesta constaba de 22 preguntas sobre demografía y actividad profesional, realización y formación en ECOPULM.
ResultadosCiento treinta y cinco socios (56,72% reumatólogos, 41,79% neumólogos) respondieron la encuesta. De ellos, el 56,30% formaban parte de una Unidad EPID en su centro. El 35,82% declararon que realizaban ECOPULM en su práctica clínica pero solo un 14,93% la realizaba en EPID, fundamentalmente con fines diagnósticos. La formación en ECOPULM de los respondedores había sido diversa en formato, contenido y auspiciadores. La gran mayoría (87,79%) consideraron que el modelo óptimo de educación en ECOPULM debería ser estandarizado y estructurado y consistir en una combinación formativa de cursos teórico-prácticos y la realización de un número mínimo de exploraciones ecográficas pulmonares supervisadas, con evaluación de competencia.
ConclusionesLa carencia actual de formación estructurada formal en ECOPULM supone una oportunidad para desarrollar programas educacionales de calidad en este campo emergente.
Interstitial lung diseases (ILD) comprise a group of diseases that share clinical, functional, and imaging manifestations, in which there is an inflammatory-fibrotic abnormality in the alveolar-interstitial structures. ILD can be classified as idiopathic and associated with pulmonary toxicants or other diseases, including systemic autoimmune diseases (SAD). ILD can affect a high percentage of patients with SAD such as antineutrophil cytoplasmic antibody-associated vasculitis, IgG4-related disease, and connective tissue diseases such as rheumatoid arthritis, systemic sclerosis, inflammatory myopathies, primary Sjögren’s syndrome, systemic lupus erythematosus or mixed connective tissue disease, in which it is one of the most important determinants of impaired quality of life and increased morbidity and mortality1,2. However, the greater availability of a much broader panel for detecting autoantibodies from immunology laboratories has made it possible to improve screening for autoimmune-derived ILD in patients with single organ disease, therefore some international societies recommend that rheumatologists and pulmonologists should collaborate in the diagnostic approach to these patients3. In this regard, an increasing number of national and international centres with interstitial lung disease units that undertake multidisciplinary work include rheumatologists as part of their teams4.
It is important to diagnose ILD early to initiate the most appropriate treatment in the early stages of irreversible structural lung damage. Functional tests are essential to detect and follow-up the disease, and DLCO (diffusing capacity of the lungs for carbon monoxide) is the measure that is affected the earliest in this type of disease5. However, low DLCO may also be due to other prevalent pulmonary diseases, such as emphysema in smokers, or the vascular involvement that accompanies some SAD such as systemic sclerosis or inflammatory myopathies, which require an imaging test. Chest X-ray has limited specificity and sensitivity and, although in suspected cases it can have a diagnostic accuracy of up to 80%6, a normal image does not exclude the diagnosis, and therefore it is mainly used to rule out other diseases associated with dyspnoea, such as infections, tumours or left heart failure7. High-resolution computed tomography (HRCT) is considered the gold standard for early and subclinical diagnosis, disease activity assessment and treatment monitoring of ILD8,9, but its use is limited by its relatively high cost and patient exposure to radiation.
Lung ultrasound (LUS) has recently emerged as a non-invasive, accessible and relatively inexpensive imaging technique that can detect lung conditions such as effusion and other pleural abnormalities, parenchymal lung consolidation and alveolar-interstitial changes of various aetiologies, including those present in ILD10–15. In numerous studies, LUS has shown high sensitivity and good concordance with HRCT in detecting and quantifying early or established lung involvement16–25. The high negative predictive value demonstrated by LUS suggests the technique could be used in the diagnosis of subclinical or early stages of ILD, as well as its consistence with HRCT, which means the latter could be spared in the follow-up and evaluation of pulmonary therapeutic response, along with clinical manifestations and respiratory functional study.
These promising applications of LUS in ILD as an efficient clinical tool currently require its learning system and technical use (appropriate equipment, scanning method and quantification of lung abnormalities) to be widely standardised so that they can be fully integrated into clinical practice and research in these diseases. There is no structured method of education and training in LUS, particularly in ILD, and very few recommendations for competency assessment and accreditation of its use in clinical practice26–28. As a first step to promote safe, appropriate, and quality use of LUS in ILD, we need to determine the current situation regarding the training and use of the procedure in our medical community.
The aim of our study was to determine education and training in LUS and its use in clinical practice and in the investigation of ILD among Spanish rheumatologists and pulmonologists.
Material and methodsStudy designThe authors, a rheumatologist (FRB) and a pulmonologist (MJRN) with expertise in ILD and attached to an ILD Unit and a rheumatologist with extensive experience in rheumatological ultrasound (EN) designed a survey addressed to members of the Spanish Society of Rheumatology (SER) (1844 members) and to members of the ILD division (1,390 members) of the Spanish Society of Pneumology and Thoracic Surgery (SEPAR). A text was drafted that included: 1) a brief explanation of the importance of the survey as a starting point for the potential development of formal and structured education offered through the SER and SEPAR for Spanish rheumatologists and pulmonologists; 2) an invitation to participate in the survey; and 3) the survey itself. The survey was presented in SurveyMonkey format.
Distribution of the surveyThe survey was advertised and distributed in August 2019 through a text advertisement and a link to the online survey inserted in three issues of the SER Newsletter (monthly electronic mailing to all SER members) and through an email to members of the SEPAR ILD division with the link to the survey. Two reminders were sent from September to December 2019. In addition, the Spanish Society of Rheumatology Ultrasound Working Group (ECOSER) invited and provided its 83 members the link to the survey by email.
Description and content of the surveyThe survey (Supplementary Material) consisted of 22 questions that covered the following: (a) demographics (two questions); (b) professional activity characteristics (four questions) on the type of work centre and medical specialty, which included the options Rheumatology, Pneumology, Paediatric Rheumatology, Paediatric Pneumology, Thoracic Surgery, Internal Medicine, Family Medicine, Physical Medicine and Rehabilitation, Intensive Care Medicine and others; c) involvement in ILD Units (two questions); d) performance of LUS in general and in ILD in particular (10 questions); e) training in LUS (four questions).
Statistical analysisThe statistical analysis was descriptive (absolute numbers and percentages) and was performed using SPSS (version 21).
ResultsDemographics and professional characteristicsA total of 135 responses were received from 74 (54.81%) women and 59 (43.70%) men; two respondents did not indicate their gender. The respondents were 76 (56.72%) specialists in Rheumatology, 56 (41.79%) specialists in Pneumology, one specialist in Internal Medicine (.75%) and 1 specialist in other specialties (.75%); one respondent did not indicate his specialty. In terms of age distribution, 12 (8.89%) respondents were under 30 years old, 44 (32.59%) between 30 and 40 years old, 31 (22.96%) between 40 and 50 years old, 38 (28.15%) between 50 and 60 years old, 9 (6.67%) between 60 and 70 years old and one (.74%) between 70 and 80 years old.
Ninety-six (71.11%) of the respondents worked in public medicine, 7 in private medicine (5.19%) and 32 (23.7%) in both public and private medicine. Most of the respondents were university-based (121; 89.63% of the respondents) and a high percentage trained residents in the speciality (101; 74.81% of the respondents).
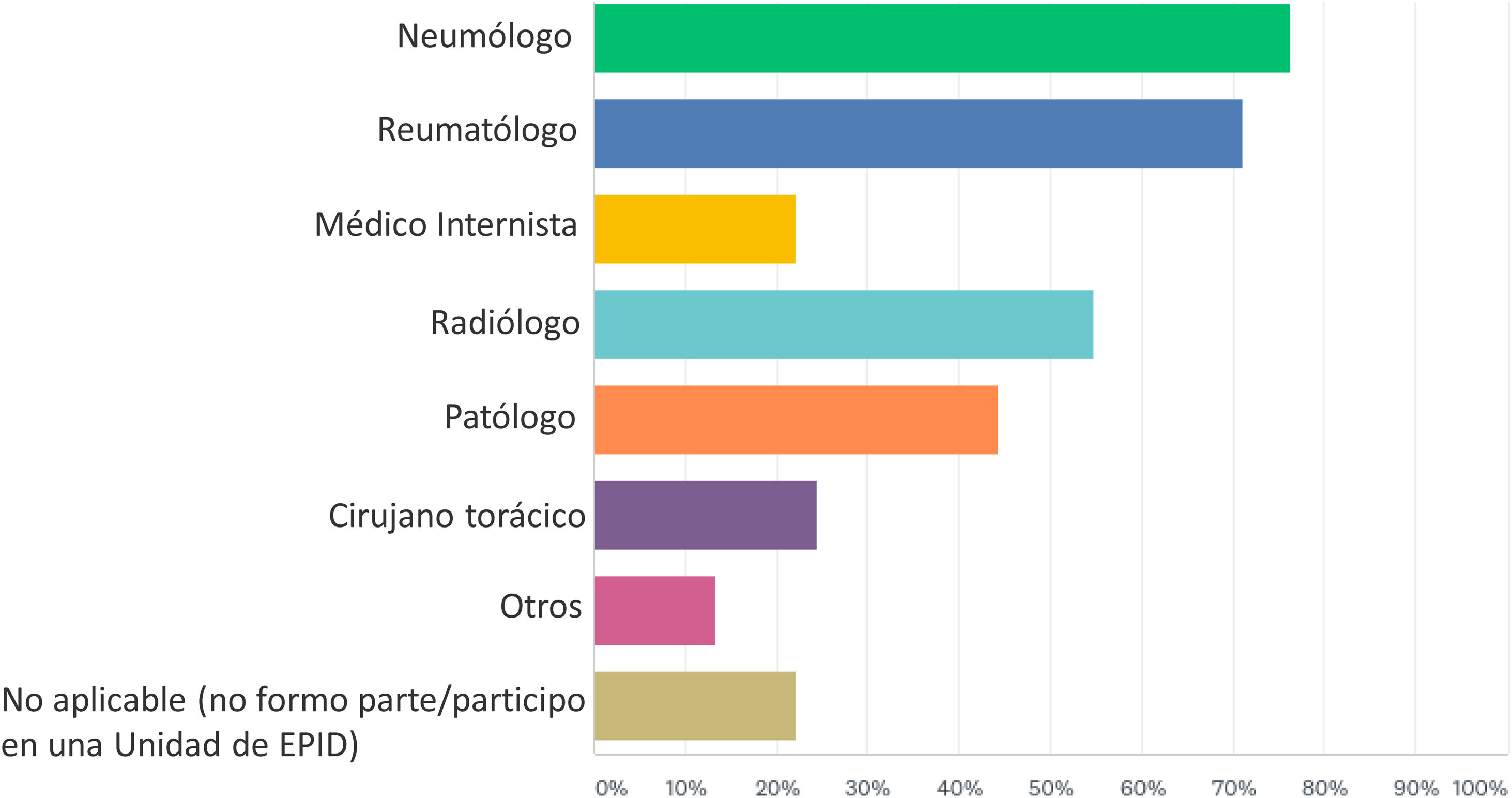
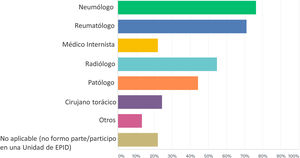
ILD unitsSeventy-six (56.30%) of the respondents were part of an ILD Unit at their workplace. The description of specialist physicians attached to ILD Units in which the respondents participated is shown Fig. 1. The most frequent medical specialties attached to ILD Units were Pneumology, Rheumatology, Radiology, and Anatomical Pathology.
Performance and use of LUSForty-eight (35.82%) respondents reported that they performed LUS in their clinical practice, while the majority, 86 (64.18%) respondents, did not perform LUS. Only 17 (12.69%) respondents performed LUS for clinical research purposes and 12 (8.96%) for basic or translational research purposes; 1 respondent did not provide information on these latter three questions.
Regarding the performance of integrated LUS in the clinic and in the inpatient area of the department/section/unit, 38 (28.57%) and 59 (44.36%) out of 133, respectively, answered in the affirmative. Only 11 (8.21%) out of 134 respondents reported that there was a LUS consultation/subunit in their department/section/unit.
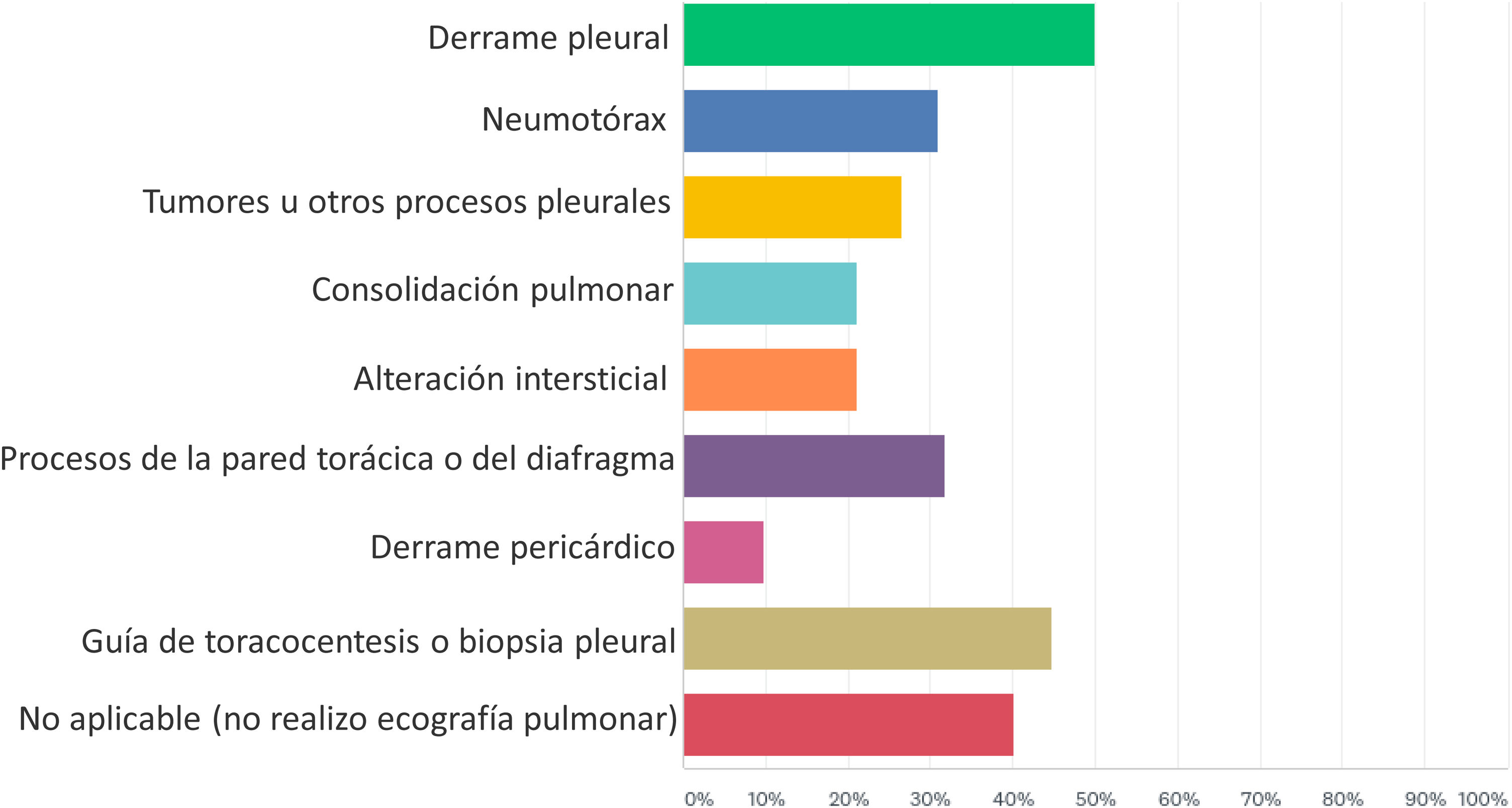
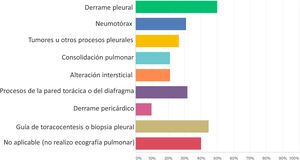
Fig. 2 shows the pathological processes in which the respondents that performed LUS applied the technique in their clinical practice. It was most frequently used to detect pleural effusion (50% of the respondents) and to guide thoracentesis or pleural biopsy (44.70% of respondents), while it was used substantially less in ILD (21.22% of the respondents). Accordingly, only 20 (14.93%) out of 134 answered yes to the question as to whether they performed LUS in ILD, for the following purposes: diagnosis (18; 13.64% of the respondents), therapeutic decision support (9; 6.82% of the respondents), monitoring of therapeutic response (7; 5.30% of the respondents), and research (13; 9.85% of the respondents). Most of the respondents who performed LUS in ILD were evaluating the presence of B-lines and pleural abnormalities as ultrasound markers of the disease process (Fig. 3).
Training in LUSTable 1 shows the LUS training methods received by the respondents. The most frequent system of education was to attend various courses (108; 81.82% of the respondents); a considerable percentage being theoretical-practical courses using healthy and diseased models.
Methods of LUS training received by the respondents.
| Response options (non-exclusive) | Respondents (n = 132) |
|---|---|
| n; % | |
| Theoretical courses | 29; 21.97% |
| Theoretical-practical courses with healthy models | 34; 25.76% |
| Theoretical-practical courses with healthy and diseased models | 45; 34.09% |
| In my own department/section/unit under expert supervision | 28; 21.21% |
| In other departments (radiodiagnosis, other) of my centre/hospital | 4; 3.03% |
| In a national centre other than where I work/worked when I trained | 12; 9.09% |
| In a centre abroad other than where I work/worked when I trained | 2; 1.52% |
| Not applicable (I have not been trained in lung ultrasound) | 44; 33.33% |
Table 2 shows the organisations or entities that organised or sponsored the LUS training received by the respondents. National or regional Scientific Societies (63; 47.37% of the respondents) and the Pharmaceutical or Technological Industry (33; 24.81% of the respondents) are the most notable.
Organisers or sponsors of the LUS training received by the respondents.
| Response options (non-exclusive) | Respondents (n = 133) |
|---|---|
| n; % | |
| National/regional scientific societies | 63; 47.37% |
| International scientific societies | 13; 9.77% |
| National universities | 3; 2.26% |
| Foreign universities | 1; .75% |
| Pharmaceutical or technological companies | 33; 24.81% |
| National or regional health system bodies | 12; 9.02% |
| Other | 13; 9.77% |
| Not applicable (I have received no training on lung ultrasound) | 44; 33.08% |
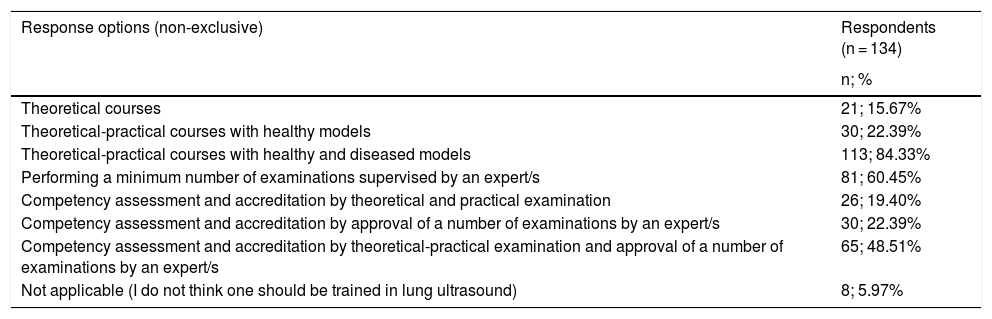
Table 3 presents the respondents' opinion on the optimal LUS training model, in terms of format, content and system of competency assessment and accreditation. Most of the respondents opted for a non-exclusive training combination of theoretical and practical courses with healthy and diseased models (113; 84.33% of the respondents) and performing a minimum number of expert-supervised lung ultrasound examinations (81; 60.45% of the respondents). In addition, most respondents supported the assessment and accreditation of competency in LUS by a theoretical-practical examination and expert endorsement of a number, to be determined, of lung ultrasound examinations performed by the trainees.
Optimal LUS training model (format, content and competency assessment and accreditation) chosen by respondents.
| Response options (non-exclusive) | Respondents (n = 134) |
|---|---|
| n; % | |
| Theoretical courses | 21; 15.67% |
| Theoretical-practical courses with healthy models | 30; 22.39% |
| Theoretical-practical courses with healthy and diseased models | 113; 84.33% |
| Performing a minimum number of examinations supervised by an expert/s | 81; 60.45% |
| Competency assessment and accreditation by theoretical and practical examination | 26; 19.40% |
| Competency assessment and accreditation by approval of a number of examinations by an expert/s | 30; 22.39% |
| Competency assessment and accreditation by theoretical-practical examination and approval of a number of examinations by an expert/s | 65; 48.51% |
| Not applicable (I do not think one should be trained in lung ultrasound) | 8; 5.97% |
Finally, the majority (115; 87.79% of respondents) stated their support for structured training in LUS, with competency assessment, for pulmonologists and rheumatologists interested in and working with lung disease processes in which this imaging method has an application
DiscussionThe ILD that occur in SAD constitute a point where care and research between Pneumology and Rheumatology meet, in which LUS could play a relevant role as an imaging tool to complement the current diagnostic, therapeutic and research approach. The reliable and efficient use of any ultrasound modality depends on the skill and experience of the examiner; therefore, the standardisation and quality of its learning and use is extremely important. Currently, training in LUS for rheumatologists and pulmonologists may be included under general specialty training in some centres in Spain and abroad, but it is not standardised and lacks competency assessment and quality certification26.
This study, based on a survey among members of the ILD division of the SER and the SEPAR, is a preliminary snapshot of the current scenario regarding use and training in LUS in both specialties and, particularly, in Spanish ILD units. Although the number of respondents was not high as a percentage of SER and SEPAR ILD division members, it does indicate a sufficiently high level of interest and motivation among Spanish rheumatologists and pulmonologists of a wide age range in the emerging field of LUS.
As might be expected, slightly more than half the survey respondents were involved in ILD units in their centres, which certainly encouraged them to collaborate in the survey. It should be noted, however, that a considerable percentage of respondents who were not involved in these multidisciplinary units also actively participated in the survey. Furthermore, the survey clearly shows how LUS is still poorly established in clinical practice and research in both specialties, particularly in the field of ILD.
Our survey showed the diversity of the current LUS training methods but showed a strong majority opinion in favour of standardising and structuring LUS training programmes, combining formal theoretical and practical training in courses and supervised practice in the clinical setting and including a competency assessment. It is very likely that the sponsoring role of Scientific Societies as providers of LUS training will soon be very welcome.
ConclusionsLUS is an emerging clinical and research tool, particularly in the synergistic setting of pulmonologists and rheumatologists who manage ILD. There is currently a lack of structured LUS training and therefore an opportunity to develop high-quality training programmes to encourage optimal use of this imaging modality in this field.
FundingNo funding was received for this study
Conflict of interestsFredeswinda Romero-Bueno has no conflict of interests to declare.
María Jesús Rodríguez-Nieto has no conflict of interests to declare.
Esperanza Naredo: Consultancy on lung ultrasound training for Bristol Myers Squibb.
We would like to thank Drs Federico Díaz-González and Carlos Sánchez-Piedra, from the Research Unit of the Spanish Society of Rheumatology (SER) for their methodological support and analysis of the results. And the SER and SEPAR for their invaluable work in distributing the Survey.
Please cite this article as: Romero-Bueno F, Rodríguez-Nieto MJ, Naredo E. Educación y uso de la ecografía pulmonar en reumatología y neumología en España: encuesta SER-SEPAR. Reumatol Clin. 2022;18:94–99.