Questionnaires to evaluate hand function are variable in the number of items, domains and diseases in which they had been previously used.
Objectivesa) To translate to Spanish and validate the Modified Score for the Assessment and Quantification of Chronic Rheumatic Affection of the Hands (m-SACRAH), and Australian/Canadian Osteoarthritis Hand Index (AUSCAN) questionnaires; b) to do a transcultural adaptation of DASHe, previously validated in Spain); and c) to compare them and the Cochin questionnaire (previously validated in México), in rheumatic patients with variable impairment of hand function.
Material and methodsm-SACRAH, AUSCAN, and Disabilities of the Arm, Shoulder and Hand (DASH) were translated/retro-translated and adapted. The final version was revised to determine content validity of m-SACRAH, AUSCAN, and DASH, plus Cochin were applied to 10 healthy subjects (pilot study) with a variable educational level and in 16 rheumatic patients with variable diagnoses and degrees of hand function impairment; all patients answered 4 questionnaires and were evaluated clinically by blinded investigators.
ResultsSeventy-six percent were women, mean age 45.7±11.4 years. Cronbach's alpha >0.90; time to answer went from 2.3±0.087 (AUSCAN) to 3.5±0.36min (DASH). There was good correlation among them (r=0.0683 AUSCAN-m-SACRAH to r=0.889 AUSCAN–DASH) and good capability for discrimination between patients with mild vs moderate to severe impairment was also demonstrated; patients with mild impairment needed less time to answer them and there were no significant differences among questionnaire scores. Patients preferred AUSCAN (10/16), Cochin (4/16), and m-SACRAH (2/16).
ConclusionThe 4 questionnaires are useful to evaluate hand function in rheumatic patients and have good discrimination capability. More patients preferred AUSCAN.
Los cuestionarios para valorar la función de las manos difieren en el número de ítems, dominios evaluados y enfermedades en las que se han utilizado.
Objetivosa) Traducir y validar al español m-SACRAH y AUSCAN; b) hacer la adaptación transcultural del DASHe, ya validado en España, y c) comparar los anteriores más el Cochin (previamente validado en México), en pacientes reumáticos y diversos grados de afección de las manos.
Material y métodosEl m-SACRAH, AUSCAN y DASH fueron traducidos/retrotraducidos y adaptados. Evaluamos validez de contenido en los 3 y, junto con el Cochin, se aplicaron a 10 sujetos sanos con escolaridad variable (estudio piloto) y a 16 pacientes reumáticos con diversos diagnósticos y grados de afección de las manos; todos respondieron los 4 cuestionarios y fueron evaluados clínicamente en forma ciega.
ResultadosEl 76% fueron mujeres, edad: 45,7±11,4 años. El alfa de Cronbach fue >0,90; el tiempo para responderlos estuvo entre 2,3±0,087 (AUSCAN) a 3,5±0,36 minutos (DASH). Hubo buena correlación de los cuestionarios entre sí (de r=0,683, AUSCAN-m-SACRAH a r=0,889 AUSCAN-DASH) y adecuada capacidad discriminatoria en pacientes con afección leve VS moderada a grave; los pacientes con afección leve tardaron menos tiempo en responderlos, no hubo diferencias significativas en las calificaciones de los cuestionarios entre sí. Los pacientes prefirieron: AUSCAN (10/16), Cochin (4/16) y m-SACRAH (2/16).
ConclusiónLos 4 cuestionarios son útiles para valorar la afección de las manos en las enfermedades reumáticas y tienen buena capacidad discriminatoria. El AUSCAN fue el que agradó a mayor número de pacientes.
Deformity of the hands is a common condition in rheumatic diseases, and pain and loss of function produce progressive difficulty in performing everyday activities, with a negative impact on the quality of life of patients. The various types of disease are related to age, sex, hand dominance, occupation, activities, physical activity, and chronic diseases. However, due to the difficulty in achieving a comprehensive evaluation of the hands of these patients, and in an attempt to make objective any symptoms and alterations in their function, different scales, and questionnaires have been created to provide a comprehensive assessment of pain, stiffness, and impact of these symptoms in activities of daily life.
The questionnaires that assess hand function published to date are: Cochin,1 m-SACRAH,2 DASH,3 which also evaluates the function of shoulder and elbow, ABILHAND,4 AUSCAN,5 the Michigan Hand Outcomes Questionnaire (MHOQ) designed for patients with various pathologies of the hand, even related to surgery,6 and Arthritis Impact Measurement Scales (AIMS),2 designed to measure the impact of disease on the health status of patients in a multidimensional way, previously translated and validated in Mexico,7 and which has even been used to evaluate the outcome of various treatments in rheumatic diseases.
The Jebsen test was designed to provide a short and objective test of hand function, evaluating activities commonly performed in everyday life, and has already been validated in rheumatic8 diseases, but requires time for its implementation (15–45min).9
Questionnaires vary in the number of items (between 12 and 78), diseases in which they have been validated (RA, OA 4), application and classification performed by the researcher. Likewise, some evaluate pain, function and stiffness, which reflects the condition of the disease on activities of daily life. DASH was recently validated in Spain,10 and Cochin and MHOQ in México11 and Colombia.12
In this study we used the Dashe, Cochin, m-SACRAH, and AUSCAN; the 4 questionnaires are similar, they all assess whether there is difficulty opening a door, 3 of them assess the ability to button a shirt, open a jar, hold and lift and open a faucet (which in Spain is known as a tap).
The objectives of this study were to translate into Spanish and validate the m-SACRAH and AUSCAN questionnaires; to perform a cultural adaptation of a previously validated version of DASH (used in Spain), plus the version of Cochin validated in Mexico and compare one another in patients with rheumatic diseases and various degrees and types of involvement of the hands.
Material and MethodsSubjectsThe translation and validation of questionnaires were performed by the following subjects: a) English/Spanish Anglophone translator, b) Participating investigators committee A (RAR, COLL, GMR, EAH, and JVM), c) Committee B: 12 people (not researchers, not doctors, and variable schooling), who were hospital workers or family members; d) healthy subjects n=10, different from committee B, but hospital workers and family, and e) patients with rheumatic disease of the hands n=16.
Questionnaires- a)
DASH. This instrument had already been translated into Spanish and validated in Spain10; starting with this release, committees A and B, led by 3 people, conducted an independent review and then met to discuss each question and its wording; during this process 5 words were replaced whose use is not common in the Spanish spoken in Mexico and, in addition, changed the presentation format for the various response options.
- b)
m-SACRAH and AUSCAN, following the methodology suggested,13–17 were first translated and then back-translated (Committee A and Anglophone translator). The committee independently reviewed the original questionnaires in English and the back-translated version first and then a joint meeting did it, and was finally approved after joint evaluation and discussion of the final versions by committees A and B.
- c)
Cochin was recently translated and validated in México11; this questionnaire was requested from the authors and the version provided by them was used in this study.
The final version of the DASH, m-SACRAH, AUSCAN, and Cochin were approved by resolution of committee A, subsequently revised and approved by committee B and, finally, became a pilot study with the healthy subjects who were asked to notify if they understood the questions and possible answers, as well as to disclose any confusion with any questions or if they had comments. According to the observations obtained in the pilot study, the committee made changes deemed necessary in the wording of the questions (face and content validity).
Questionnaires were applied on a normal day at the rheumatology clinic and patients who agreed to participate in the study through written informed consent were evaluated by the rheumatologist responsible and subsequently received a booklet, responding alone, with the help of a relative or interviewer if you so required. The booklet consisted of the 4 questionnaires (m-SACRAH, AUSCAN, Cochin, and DASH) in variable order; we recorded the time required for patients to answer each one and, finally, we asked which questionnaire they preferred.
The reliability or consistency was measured with Cronbach's alpha in the 4 questionnaires. The stability was determined in some patients without clinical changes in which we applied one of the four questionnaires, 3 days after the first.
Clinical EvaluationThe variables analyzed in the patient group were: age, sex, hand dominance, diagnosis, number of tender, swollen joints, and limited mobility. The rheumatologist assessment was a pre-response to questionnaires and included 2 visual analog scales from 0 (best) to 10cm (worst possible), the first referring to the function of the hands and the second to the patient overall (construct validity).
As part of the analysis, we evaluated the discriminatory ability of the questionnaires between 2 groups with condition of the hand, mild affection (assessment of hand function by VAS: 0–4cm) and moderate to severe disease (VAS hand function 5–10cm).
Statistical AnalysisInitially, all subjects (healthy and patients) were included in a group with varying disorders in hand function, regardless of diagnosis they had. We assessed whether a greater clinical dysfunction of the hands was related to demographic and clinical variables, and higher scores on the various function questionnaires using t test, linear correlation and Cronbach's alpha. The score of the questionnaires on the day of the visit was compared with the same score after 3 days (test-retest), using intraclass correlation coefficient.
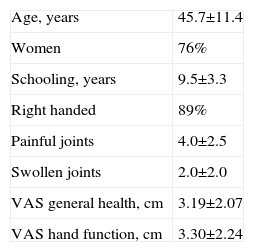
ResultsCharacteristics of SubjectsWe included 10 healthy subjects and 16 patients with rheumatic diseases and varying degrees of involvement of the hands (3 with osteoarthritis of the hands,18 5 with rheumatoid arthritis,19 4 with gout20 and 4 with systemic sclerosis21); data shown in Table 1.
General Characteristics of Participants (Healthy n=10 and Rheumatic Patients n=16).
| Age, years | 45.7±11.4 |
| Women | 76% |
| Schooling, years | 9.5±3.3 |
| Right handed | 89% |
| Painful joints | 4.0±2.5 |
| Swollen joints | 2.0±2.0 |
| VAS general health, cm | 3.19±2.07 |
| VAS hand function, cm | 3.30±2.24 |
X±SD except when specified.
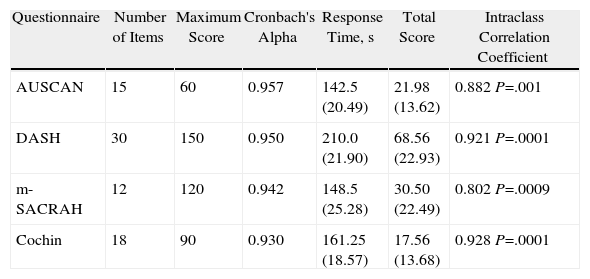
The 4 questionnaires were similar for the assessment of rheumatic patients; the general data of each are found in Table 2.
Characteristics of AUSCAN, DASH, m-SACRAH, and Cochin Questionnaires.
| Questionnaire | Number of Items | Maximum Score | Cronbach's Alpha | Response Time, s | Total Score | Intraclass Correlation Coefficient |
| AUSCAN | 15 | 60 | 0.957 | 142.5 (20.49) | 21.98 (13.62) | 0.882 P=.001 |
| DASH | 30 | 150 | 0.950 | 210.0 (21.90) | 68.56 (22.93) | 0.921 P=.0001 |
| m-SACRAH | 12 | 120 | 0.942 | 148.5 (25.28) | 30.50 (22.49) | 0.802 P=.0009 |
| Cochin | 18 | 90 | 0.930 | 161.25 (18.57) | 17.56 (13.68) | 0.928 P=.0001 |
Mean values, standard deviation shown in parenthesis.
Response time and total score of included subjects (healthy and rheumatic patients).
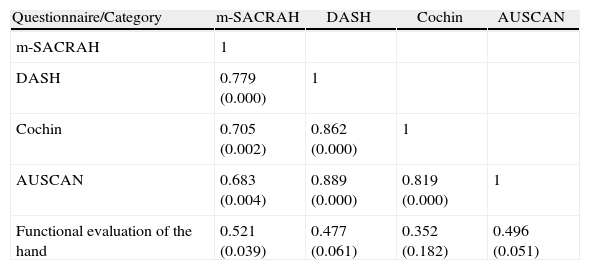
There was good correlation of the 4 questionnaires with each other (Table 3) and although smaller in power, with hand function (VAS) conducted by the physician, and this, in turn, was related to the general health VAS status (r=0.649, P=.006) and number of swollen joints (r=0.585, P=.017). All questionnaires had a tendency to show lower scores, although this may be secondary to most of the patients having mild to moderate functional impairment.
Correlation Between Questionnaires and Functional Evaluation of the Hands.
| Questionnaire/Category | m-SACRAH | DASH | Cochin | AUSCAN |
| m-SACRAH | 1 | |||
| DASH | 0.779 (0.000) | 1 | ||
| Cochin | 0.705 (0.002) | 0.862 (0.000) | 1 | |
| AUSCAN | 0.683 (0.004) | 0.889 (0.000) | 0.819 (0.000) | 1 |
| Functional evaluation of the hand | 0.521 (0.039) | 0.477 (0.061) | 0.352 (0.182) | 0.496 (0.051) |
Number refers to r value; P is shown in parenthesis.
Functional evaluation of the hand evaluated by a physician using a visual analog scale.
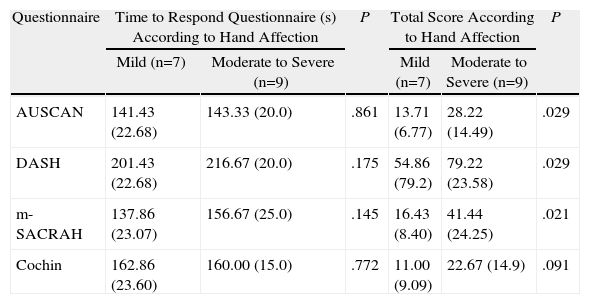
When patients were divided into 2 groups according to the hand function VAS, no significant differences between groups were seen in terms of age and education (45±12 years vs 43±7 and 9 years±4 vs 8.7±3, respectively), or in the required time to complete the questionnaires.
However, the total score was different in patients with mild vs moderate to severe disease for the AUSCAN, m-SACRAH, and DASH questionnaires; the difference was not significant for Cochin (Table 4).
Comparison of Questionnaires According to the Degree of Hand Affection.
| Questionnaire | Time to Respond Questionnaire (s) According to Hand Affection | P | Total Score According to Hand Affection | P | ||
| Mild (n=7) | Moderate to Severe (n=9) | Mild (n=7) | Moderate to Severe (n=9) | |||
| AUSCAN | 141.43 (22.68) | 143.33 (20.0) | .861 | 13.71 (6.77) | 28.22 (14.49) | .029 |
| DASH | 201.43 (22.68) | 216.67 (20.0) | .175 | 54.86 (79.2) | 79.22 (23.58) | .029 |
| m-SACRAH | 137.86 (23.07) | 156.67 (25.0) | .145 | 16.43 (8.40) | 41.44 (24.25) | .021 |
| Cochin | 162.86 (23.60) | 160.00 (15.0) | .772 | 11.00 (9.09) | 22.67 (14.9) | .091 |
Mean values, standard deviation shown in parenthesis.
Functional evaluation of the hand evaluated by a physician using a visual analog scale.
After answering them, we asked patients to indicate which of the 4 was their favorite: 62.5% (10/16) preferred AUSCAN, 25% (4/16) Cochin, and 12.5% (2/16) the m-SACRAH; no patients preferred the DASH.
DiscussionThe 4 questionnaires showed validity for assessing the function of the hands of Mexican patients with nodal osteoarthritis, rheumatoid arthritis, gout and systemic sclerosis. The time and trouble to answer them was similar between them. The degree of hand function was related to the assessment of the treating physician.
Although patients with more affection of the hands required more time to answer them, the differences were not significant. The scores of Cochin in patients with greater hand dysfunction was nearly double those of lesser condition, but this difference was not significant, which if compared with the other questionnaires shows the difference in score between both groups as higher.
A higher percentage of patients preferred AUSCAN and none the DASH, probably because in the latter, the function of the elbows and shoulders is also assessed, is longer and requires more time to answer.
All patients included in this study were seen at the rheumatology clinic and none had severe functional disability; the usefulness of questionnaires in patients with severe disability is not known, because these patients generally are not included in studies.
In daily practice, the use of AUSCAN, mSACRAH, Cochin, and DASH in versions validated for Mexico provides a useful assessment of the functional aspect of the hands in patients with rheumatic diseases, as demonstrated with other versions of these questionnaires, especially in patients with rheumatoid arthritis and osteoarthritis.22,23
In conclusion, we compared conceptually equivalent questionnaires in Spanish to their original versions, and according to the results, the AUSCAN questionnaire outperformed others in rheumatic patients, so it could be a useful tool for everyday use in patient assessment.
Ethical disclosuresProtection of human and animal subjects. The authors declare that the procedures followed were in accordance with the regulations of the responsible Clinical Research Ethics Committee and in accordance with those of the World Medical Association and the Helsinki Declaration.
Confidentiality of Data. The authors declare that no patient data appears in this article.
Right to privacy and informed consent. The authors have obtained the informed consent of the patients and /or subjects mentioned in the article. The author for correspondence is in possession of this document.
Conflict of InterestThe authors declare no conflict of interest.
We appreciate the collaboration of Dr. José Alvarez Nemegyei for his validated Mexican version of the Cochin; to professor Margaret Gunzburger for her participation in the questionnaire translation process, and to Ma. Eugenia Sanchez Girard for her role as research assistant.
Please cite this article as: Arreguín Reyes R, et al. Evaluación de la función de la mano en las enfermedades reumáticas. Validación y utilidad de los cuestionarios AUSCAN, m-SACRAH, DASH y Cochin en Español. Reumatol Clin. 2012. http://dx.doi.org/10.1016/j.reuma.2012.03.005