A high serum level of creatine kinase (CK) is a common reason for referring to medical specialities. Myopathies are one of the causes of elevated levels of CK. McArdle disease is the most common disorder of skeletal muscle carbohydrate metabolism. The cases are presented on 2 patients who were referred to our medical consultation to study the cause of their increased CK levels: a 72-year-old asymptomatic man with high levels of CK detected by chance in a routine analysis, and a 30-year-old woman with very few symptoms, apart from slight muscle pain and tiredness. Electromyography was normal in both patients. There was null activity of myophosphorylase in muscle biopsy of both cases, so a diagnosis of McArdle disease was made.
El estudio del incremento de la creatinina fosfoquinasa (CPK) constituye un motivo de consulta frecuente en diversas especialidades médicas. Entre las enfermedades que cursan con CPK alta se encuentran las miopatías metabólicas siendo la enfermedad de McArdle la glucogenosis muscular más frecuente. Presentamos 2 casos clínicos de pacientes derivados a nuestro servicio de reumatología para estudio de CPK elevada cuyo diagnóstico final fue enfermedad de McArdle: un hombre de 72 años, asintomático desde el punto de vista muscular, en el que se objetivó de manera casual elevación importante de CPK en una analítica de rutina y una mujer de 30 años con síntomas musculares muy leves. El electromiograma (EMG) fue normal en ambos pacientes. En ninguno de los 2 casos existía actividad de la miofosforilasa en la biopsia muscular, siendo diagnosticados de enfermedad de McArdle.
Increases in the creatine kinase (CK) level are frequent reasons for consulting specialists in rheumatology, neurology and internal medicine. Metabolic myopathies are diagnosed in 41% of the cases of asymptomatic or nearly asymptomatic high serum CK levels.1
McArdle disease (also known as glycogen storage disease type V) is a metabolic myopathy caused by deficiency of the enzyme myophosporylase.2 The clinical characteristics are exercise intolerance, poor stamina and painful muscle cramps; in addition, myoglobinuria, rhabdomyolysis and acute renal failure can develop in 50% of the cases.3,4 The diagnosis is based on an increase in glycogen content and decrease in enzyme activity observed in a muscle biopsy.5
Although there is no specific treatment for this disease at the present time, it is possible to improve the quality of life of the patients and prevent rhabdomyolysis and renal failure with the proper diet and by controlling physical activity.3
We present the case reports of 2 patients, studied in our department because of elevated CK levels, who were ultimately diagnosed with McArdle disease.
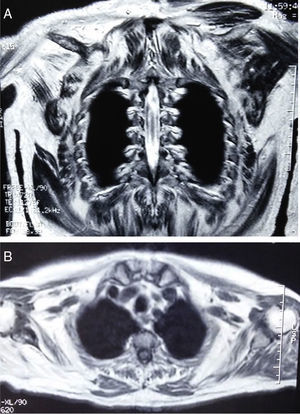
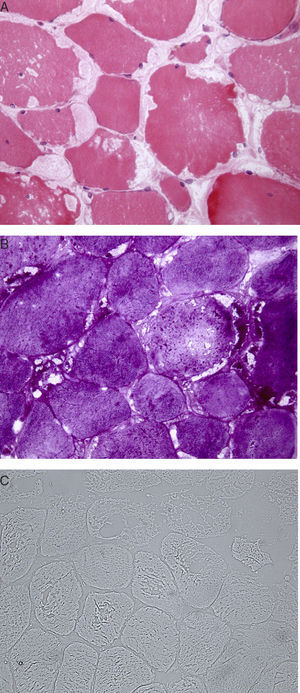
Clinical ObservationsCase report no. 1. The patient was a 72-year-old man with Paget's disease who was being followed in periodic visits to the rheumatology outpatient clinic. Although he had no symptoms of muscle involvement, an elevated CK level was an incidental finding in a routine analysis. A review of previous analyses revealed that it had been rising progressively, and had reached values of 5000IU/L. He also had slightly elevated aldolase levels, with no changes in liver enzymes or thyroid status, and negative autoantibody tests. Physical examination showed normal muscle balance. An electromyogram (EMG) revealed no evidence of myopathy. Magnetic resonance imaging (MRI) of the shoulder girdle showed bilateral atrophy and fatty replacement in several muscles (Fig. 1A and B). It also revealed changes in the deltoid muscle, which was biopsied. The pathological study disclosed the presence of subsarcolemmal glycogen-containing vacuoles (Fig. 2A and B). Histochemical analysis demonstrated a total absence of myophosphorylase, a finding compatible with McArdle disease. In the genetic study, a homozygous Y733X nonsense mutation was observed in exon 18 of the PYGM gene. The patient was referred to the endocrinology-nutrition department.
Case Report no. 2. This patient was a 30-year-old woman with nothing noteworthy in her clinical history. She had been referred to our department from primary care for the study of persistent CK elevation (peak value: 4000IU/L). She complained of asthenia and the feeling of a slight loss of strength in her lower limbs following strenuous exercise. On physical examination, her muscle balance was normal. There was no EMG evidence of myopathy. Laboratory tests showed an elevated CK level and normal liver function, aldolase and lactate dehydrogenase levels, and autoantibody profile. The results of the determination of thyroid hormone levels led to a diagnosis of subclinical hypothyroidism, which did not require treatment. Given that CK continued to be elevated in follow-up analyses, and based on her report of weakness in her lower limbs following strenuous physical exercise, the decision was made to perform a biopsy of the quadriceps muscle. The results revealed the presence, in the sarcolemma, of multiple clear vacuoles containing eosinophilic granules. Moreover, the histochemical study demonstrated the absence of activity of the enzyme myophosphorylase, a finding that is compatible with McArdle disease (Fig. 2C). A genetic study was not carried out. This patient was also referred to the endocrinology-nutrition department.
DiscussionMcArdle disease, described for the first time in 1951 by Dr. Brian McArdle,6–8 is the most frequently found muscle glycogenosis and one of the most common genetic myopathies.3 Nevertheless, given its low incidence, it is included in the group of “rare diseases” (up to 2012, there were 239 cases registered in Spain).9 Its prevalence in Spain is estimated to be 1/167000.8 It is caused by autosomal recessive mutations in the myophosphorylase gene (PYGM) located on chromosome 11.3,8,10 This enzyme has a role in the first step in skeletal muscle glycogen metabolism, and is necessary for the conversion of this polysaccharide into glucose-1-phosphate. Thus, myophosphorylase deficiency makes it impossible to obtain energy from the glycogen stores in muscle.3,8
The clinical signs and symptoms include exercise intolerance with myalgia, stiffness and cramps.3 Strenuous exertion can be followed by myoglobinuria and acute renal failure, the former in up to half of the cases.2,3,5,8 It has a pathognomonic characteristic known as the “second wind” phenomenon, in which the patient acquires greater tolerance to physical exercise after the first 10min have elapsed.2,3,11 The symptoms usually appear during adolescence or early adulthood (the patient in our second case began to notice fatigue and myalgia at the age of 30 years).2,3,7,8 Nevertheless, there is considerable heterogeneity in the severity of the presenting signs and symptoms, ranging from congenital myopathy, in which there are nearly no symptoms, as occurred in our first patient, to a variant with a fatal outcome that appears during childhood.2,3,5 Cases of late onset McArdle disease involving muscle atrophy and predominantly proximal weakness have also been reported.3,7,12,13
The characteristic laboratory findings are elevated CK levels (even during intervals between peaks in disease activity).2,3,5 The EMG can be normal or show evidence of myopathy.5 If the ischemic forearm exercise test is performed, the curve observed for lactic acid is flat, indicative of failure of the breakdown of glycogen to lactate.5,14 Regarding imaging studies, both computed tomography and MRI can demonstrate atrophy and fatty replacement in muscle, especially in cases involving late onset, as in our first patient.12,15 The definitive diagnosis is based on muscle biopsy and the observation of the absence of myophosphorylase in a subsequent immunohistochemical analysis.3,5 The histological findings are characterized by the presence of subsarcolemmal vacuoles containing glycogen, which can be examined using periodic acid Schiff (PAS) staining.3 The diagnosis should be corroborated by a genetic study.3,5,6,8
Treatment is based on the control of physical activity and dietary measures.3,16 The diet should be rich in carbohydrates (65%) and low in fat (20%), and the patients are informed of the need to consume foods containing simple carbohydrates 5min before exercising. Moderate regular physical activity has been shown to increase exercise tolerance in these patients.3,17,18
We conclude that, in cases of elevated CK levels, regardless of patient age and the severity of the muscle damage, the differential diagnosis should include McArdle disease.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of InterestThe authors declare they have no conflicts of interest.
We thank Dr. Carmen Navarro for her contribution to this study.
Please cite this article as: Diez Morrondo C, Pantoja Zarza L, San Millán Tejado B. Enfermedad de McArdle: presentación de 2 casos clínicos. Reumatol Clin. 2016;12:161–163.