More than 50% of the patients with primary Sjögren's syndrome (pSS) have extraglandular manifestations, and pulmonary manifestations are observed in 10% of this patient population.1 In 50% of the patients, the involvement is subclinical; however, symptomatic lung disease occurs in 10%. Interstitial lung disease is the most common disorder.2–4 Cystic lung disease is reported much less frequently. This complication is characterized by foci of reduced lung density that have well-defined, thin walls (wall thickness less than 4mm) and a diameter at their largest point of 1cm.5 We report the cases of 3 patients with cystic lung disease associated with pSS. All of them met the 2002 American-European classification criteria for the diagnosis of Sjögren's syndrome (SS).6
The 3 pSS patients were diagnosed, by means of high-resolution computed tomography (HRCT) of the thorax, as having associated cystic lung disease, together with centrilobular pulmonary emphysema, predominantly in the upper lobes in 2 of the 3 patients. There was no evidence of association with other lung diseases. We did not perform a histological examination, a study that had been described in other reports published up to that time.
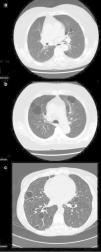
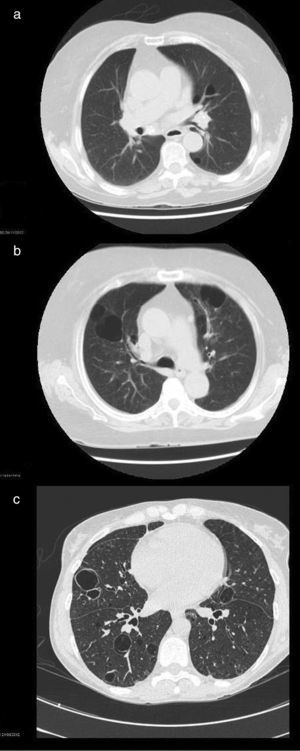
Patient no. 1The first patient was a 74-year-old woman with an 18-year history of pSS. She presented with extraglandular manifestations, such as the vasculitis, polyarteritis nodosa. The most notable laboratory findings were hypergammaglobulinemia and anti-Ro and anti-La antibodies. She was being treated with oral azathioprine at a dose of 50mg/day. During follow-up she developed functional dyspnea grade II. The results of respiratory function tests were normal, and HRCT of the thorax revealed centrilobular pulmonary emphysema, predominantly in upper lobes, and images of thin-walled cysts predominantly in lower lobes (Fig. 1a). The same treatment was maintained, and there were no changes in the pulmonary lesions in subsequent visits.
High-resolution computed tomographic study: (a) image showing centrilobular emphysema and cysts; (b) bilateral cystic lung lesions, some with septa in their interior, traction bronchiectasis in both lower lobes, areas of centrilobular and paraseptal emphysema and zones of fibrotic tissue; and (c) image showing multiple bilateral thin-walled pulmonary cysts.
This patient was a 46-year-old woman with a 20-year history of pSS and diagnosed with IgA kappa myeloma. Her extraglandular manifestations were anemia and leukocytoclastic vasculitis, as well as polyarteritis and parotid gland enlargement. She was being treated with hydroxychloroquine (HCQ) 200mg/day, oral pilocarpine 15mg/day and rituximab every 6 months. Laboratory tests revealed elevated acute-phase reactant levels, anemia and lymphopenia, as well as hypergammaglobulinemia with an elevated IgG level and a monoclonal IgA kappa component. Tests for rheumatoid factor (RF), as well as anti-Ro and anti-La antibodies, were positive. During follow-up, she developed dyspnea on heavy exertion. Respiratory function tests revealed a mild restrictive ventilatory defect with a slightly reduced diffusing capacity of the lung for carbon monoxide (DLCO), and HRCT of the thorax showed bilateral cystic lung lesions—some with septa in their interior—predominantly in the bases, traction bronchiectasis in both lower lobes, areas of centrilobular and paraseptal emphysema in upper lobes, together with linear opacities and areas of fibrotic tissue in both lung bases (Fig. 1b). No modifications were made in her treatment, and there were no changes in the cystic lesions in subsequent visits.
Patient no. 3The third patient was a 78-year-old woman with a 15-year history of pSS who was also diagnosed with cutaneous amyloidosis. The manifestations were predominantly glandular (xerostomia and xerophthalmia), and there was no extraglandular involvement. She was receiving oral HCQ 200mg/day. The laboratory tests revealed elevated acute-phase reactant levels, polyclonal hypergammaglobulinemia with an elevated IgG level, and positive tests for RF and anti-Ro and anti-La antibodies. During follow-up, she reported dyspnea on moderate exertion and, thus, underwent respiratory function tests, which revealed a mild restrictive ventilatory defect, with a severe reduction in DLCO. Thoracic HRCT (Fig. 1c) showed multiple bilateral thin-walled pulmonary cysts, with no evidence of nodules or changes in the air space. The findings were the same in subsequent visits.
Cystic lung disease is an uncommon condition that is rarely associated with an autoimmune inflammatory disease. The prevalence of pulmonary cysts in pSS ranges between 12% and 30% of the overall group of pulmonary manifestations. They can be found alone or in association with areas of ground glass attenuation.7–9 Two hypotheses have been proposed for the formation of these pulmonary cysts, both of which involve the infiltration of immunoglobulin-producing lymphocytes: a valve mechanism that causes cystic changes in the dilated alveolar region as a result of chronic inflammation and lymphoplasmocytic infiltration in peripheral regions; and the destruction of the alveolar wall, in which the direct lymphoplasmocytic infiltration of the alveoli destructs the alveolar structures and creates cystic areas. The most probable scenario involves both mechanisms.7,8,10 There have been few reports of pulmonary cysts associated with pSS, and the majority of the cases are found to be concomitant with other diseases, such as lymphoid interstitial pneumonitis, lymphoproliferative processes and pulmonary amyloidosis.11,12 One common characteristic finding is the high erythrocyte sedimentation rate with hypergammaglobulinemia observed in all 3 cases, a direct reflection of the lymphocyte hyperactivity associated with the disease. Compared with the cases in the literature, our patients do not differ with respect to the characteristic radiological findings,10 although there have been reports of a significant association between seropositivity for anti-SSB/La and clonal lymphoproliferative disorders that we have not identified in our patients.7
Please cite this article as: Masegosa-Casanova S, Riveros-Frutos A, Sanint J, Olivé A. Quistes pulmonares asociados a síndrome de Sjögren primario. Reumatol Clin. 2016;12:116–118.