Imaging may be useful for monitoring response to therapy. Within the OMERACT proposal for the core set domains for outcome measures in chronic gout, serum urate levels, recurrence of gouty flares, tophus regression, and joint damage imaging have been included, among other proposed issues.
ObjectivesTo perform a systematic literature review of the usefulness of magnetic resonance imaging (MRI) and ultrasound (US) on assessment of treatment response in patients with gout.
MethodsMEDLINE, EMBASE, Cochrane Library (up to February 2012), and abstracts presented at the 2010 and 2011 meetings of the American College of Rheumatology and European League Against Rheumatism were searched for treatment studies of any duration and therapeutic options, examining the ability of MRI/US to assess treatment response in gouty patients. Meta-analyses, systematic reviews, randomized clinical trials, cohort and case-control studies and validation studies were included. Quality was appraised using validated scales.
ResultsThere were only 3 US published studies in the literature that analyzed US utility on assessment of response to treatment in patients with gout. All of them were prospective case studies with a small number of patients and they were reviewed in a detailed manner. A total of 36 patients with gout were examined with US. All of them had a baseline serum urate >6mg/dL. US features of gout (double contour sign, hyperechoic spots in synovial fluid, hyperechoic cloudy areas, tophus diameter and volume) achieved significant reduction in patients who reached the objective of uricemia ≤6mg/dL in all the studies; however, patients in whom levels did not drop below 6mg/dL had no change of US features of gout. Other parameters evaluated in one study included ESR, CRP, number of tender joints (TRN), number of swollen joints, and pain score (SP). All of them decreased with uricemia reduction, but only TRN and SP were statistically significant. No data were found on the value of MRI on treatment response assessment in patients with gout.
ConclusionsThe improvement in ultrasound features shows concurrent validity with uric acid reduction. According to the published evidence, US can be a useful tool for monitoring treatment of gouty patients, although more research is needed. The value of MRI on treatment response assessment in patients with gout remains to be determined.
Evaluar sistemáticamente la evidencia publicada en relación con la utilidad de la ecografía y de la resonancia magnética nuclear (RMN) en la evaluación de la respuesta al tratamiento en pacientes con artritis por depósito de cristales de urato monosódico.
MétodosRevisión sistemática. Se definió una estrategia de búsqueda bibliográfica sensible en MEDLINE (desde 1960), EMBASE (desde 1980) y Cochrane Library (Central) y búsqueda manual en lo publicado en congresos internacionales (EULAR, ACR), seleccionando estudios que evaluasen la respuesta al tratamiento de los pacientes con gota (AINE, hipouricemiantes, etc., independientemente del tipo, de la dosis y de la duración del tratamiento e independientemente del criterio seleccionado para el diagnóstico, el tipo y el número de articulaciones afectadas) mediante pruebas de imagen (ecografía y/o RMN). Se seleccionaron metaanálisis, revisiones sistemáticas, ensayos clínicos, cohortes de calidad y estudios de validación.
ResultadosLos estudios incluidos fueron 3 series de casos, de diseño prospectivo, en los que se analizó un total de 36 pacientes con artritis gotosa y en los que se evaluó la respuesta al tratamiento hipouricemiante mediante ecografía. No hubo ningún estudio que analizara la respuesta al tratamiento hipouricemiante con RMN. Todos los pacientes incluidos tenían valores basales de uricemia por encima de las recomendaciones EULAR. La mejoría en los hallazgos ecográficos se correlacionó con la reducción de los valores séricos de ácido úrico en todos los estudios.
ConclusionesEl uso de la ecografía parece útil en la evaluación de la respuesta al tratamiento hipouricemiante en los pacientes con artritis gotosa (nivel de evidencia 3a; grado de recomendación B). Actualmente no se dispone de ninguna evidencia para analizar la utilidad de la RMN en la evaluación del tratamiento de los pacientes con artritis gotosa.
Gout is caused by the formation and deposition of monosodium urate crystals (MSU) in joints, or by forming usually extra-articular nodules called tophi. For the crystals to form, uric acid concentrations above 6.8mg/dL in blood are required, which is the saturation point for1,2 MSU .Once formed, the crystals remain in the joint cavity indefinitely while hyperuricemia, persists and over time the deposit increases and affecting more joints. However, the deposition of crystals may be reversed and, if the concentration of uric acid decreases, the crystals are dissolved and gout can be considered cured.3,4
As recommended by the EULAR working group for the treatment of gout, hypouricemic treatment goal is to promote the dissolution of the crystals and prevent their formation; this is achieved by maintaining a serum uric acid concentration below the saturation point for MSU: ≤6mg/dL (360≤mol/L).5,6
Various trials and cohort studies have shown an inverse correlation between serum uric acid levels during treatment and the speed with which the subcutaneous tophi7,8 are reduced. The reduction of uric acid below 6mg/dL is associated with not only a progressive reduction of subcutaneous tophi, but also reducing the size of the deep tophi, not accessible to physical examination and yet others detectable by imaging techniques such as ultrasound and magnetic resonance imaging (MRI).8 Since these techniques are valid for determining the size of tophi and their location, it is of great interest to know their usefulness in evaluating the effectiveness of hypouricemic treatment, which helps decide not only when to use hipouricemic agents in patients with gout but also when to intensify treatment or whether it could be reduced or even suspended.
Therefore, the objective of this systematic review is to analyze the published evidence on the usefulness of ultrasound and MRI in the evaluation of treatment response in patients with gout.
Materials and MethodsWe selected studies that evaluated the response to treatment of patients with gout (NSAIDs hipouricemic agents, etc., regardless of type, dose and duration of treatment) using imaging (ultrasound and/or MRI) in patients with gouty arthritis (selected regardless of the criteria for diagnosis, type and number of joints affected). We selected articles that measured correlation coefficients, sensitivity to change, changes after treatment using radiological and clinical variables (number of attacks, number of swollen joints, uric acid levels, number of tophi, disappearance of tophi, quality of life and laboratory data), variables related to safety, etc. Regarding the design of the studies, meta-analyses, systematic reviews, clinical trials, cohort and validation studies were included. Basic science articles and animal studies were excluded.
We searched the following electronic databases until February 2012: MEDLINE (from 1960), EMBASE (from 1980) and the Cochrane Library (Central). We searched both MeSH terms and free text formats. No limits were set on the publication date. In addition, we searched abstracts sent to ACR and EULAR meetings in 2010 and 2011. Finally, a manual search for articles finally included was performed. The specific search strategy is detailed in the supplementary material (see Appendix annex).
A single reviewer (VV) analyzed the articles resulting from the search as well as the detailed analysis of the items included. The first search result was filtered by title and abstract or entire article in case they were not summarized by 20min maximum duration sessions. If in doubt, a second reviewer (EL) resolved the discrepancy. After this process, the remaining items were analyzed in detail (VV). Again, if in doubt, EL would solve it. Finally, a manual search with references selected for detailed analysis was performed. All Internet references were retrieved and placed in the EndNote software to facilitate management.
To assess the methodological quality of included studies we used: (a) for clinical trials, the Jadad scale (1–5; considering good quality studies as Jadad 3–5), and (b) for cohort studies, the Oxford quality scale.
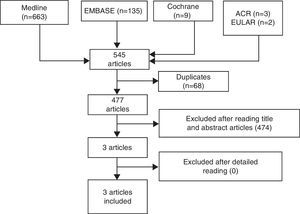
ResultsThe search results are detailed in Fig. 1.
Overview of Articles Included3 studies9–11 were eligible for inclusion, all small prospective case series, which included a total of 36 patients with gouty arthritis (detection of uric acid crystals in the synovial fluid with polarized light microscope) and whose response to hypouricemic treatment was assessed by ultrasound. Uric acid blood concentration determinations were performed every 3 months, and one study11 further recorded: pain by visual analog scale (VAS), global assessment of the disease by the patient (VAS), number of tender and swollen joints, excretion of uric acid, CRP and ESR. Hypouricemic treatment was adjusted based on EULAR recommendations (blood uric acid ≤6mg/dL). As for the ultrasound variables analyzed, one study evaluated the change in the size of tophi,9 another measured persistence/decrease or disappearance of the ultrasound “double contour” (deposits of uric acid crystals in the hyaline cartilage) based on serum uric acid levels10 and, in the latter, in addition to evaluating the sonographic “double contour” sign, the Doppler signal was analyzed, as was the presence or absence of hyperechoic spots in the synovial fluid and the presence or not of hyperechoic “cottony images”11 (fig. 1).
The main features of the articles included in this systematic review are shown in Table 1.
Table of Evidence.
| Study | Population | Intervention | Outcome measures | Commentsa |
| Perez-Ruiz, 20079 (1)12 monthsProspective case series | n=22EC: severe gout, acute inflammation | USMRI (gold standard) | Apparent validityReliability (inter- and intraobserver correlation, multiple regression, intraclass correlation)Sensitivity to changeChanges in the size and volume of tophiAdverse Events | Oxford 3A |
| Thiele, 201010 (2)7–18 monthsProspective case series | n=5 patients | US | Apparent validityReliability (inter- and intraobserver correlation)Sensitivity to changeChanges in uric acid deposits in the hyaline cartilageAdverse Events | Oxford 3b |
| Peiteado et al., 201011 (3)12 monthsProspective case series | n=17 patientsEC: patients with other rheumatic diseases | US | Apparent validityReliability (inter-and intraobserver correlation)Sensitivity to changeChanges in uric acid deposits in the hyaline cartilage, synovial fluid deposits, Doppler signal, ESR, CRP, NSJ, NTJ, VAS pain and GPEAdverse Events | Oxford 3b |
EC: exclusion criteria; IC: inclusion criteria; VAS: visual analog scale; NTJ: number of painful joints; NSJ: number of swollen joints; CRP: C-reactive protein: MRI: magnetic resonance imaging; U.S.: musculoskeletal ultrasound; GPE: global evaluation of disease by the patient.
All patients had baseline serum uric acid above the EULAR recommendations (>6mg/dL in the study by Thiele and Schlesinger,10 14mg/dl in the Perez-Ruiz et al. study9 and 17mg/dL in the Peiteado et al. paper11), and therefore, in the 2 studies in which the ultrasound “double contour” sign10,11 was assessed, this was present at baseline in all patients. In both studies, the improvement in ultrasound findings correlated with the reduction of serum uric acid. The double contour sign is significantly reduced and even disappeared in patients who achieved the target (uricemia ≤6mg/dL), but no changes occurred in patients whose serum uric acid values did not decrease below this figure. Other activity parameters evaluated in the study by Peiteado et al.,11 decreased with good control of uric acid levels but did not reach statistical significance: ESR, CRP, number of swollen joints and doppler ultrasound signal.
In the study by Perez-Ruiz et al.,9 wherein the changes detected by ultrasound regarding the size and volume of tophi were analyzed, it was observed that most of the tophi had changes in the maximum diameter or volume, and this occurred in patients who had an adequate control of serum uric acid. Thus, in patients with uric acid below 6mg/dL, 68% had a reduction of tophi below the minimum detectable difference, while only 10% of patients with tophi and a uric concentration above 6mg/dL had a significant reduction. In addition, there was an inverse correlation between the reduction of the maximum diameter, transverse diameter and mean tophi volume and serum uric acid concentration during hypouricemic treatment.
No study reported any adverse event in relation to the use of ultrasound.
Excluded StudiesThe exclusion table is not shown, since all selected articles were included in detail.
DiscussionIn this systematic review of the literature we analyzed the value of ultrasound and MRI in the evaluation of treatment response in patients with gouty arthritis. To achieve this goal, we conducted an extensive search on the main bibliographic databases available, as described in the Materials and Methods section. After detailed analysis of the selected studies, only three studies were included that examined the utility of ultrasound to evaluate the response to treatment, 2 published in international journals and one9,10 presented solely as a poster at EULAR.11 All were case series conducted at a single center with a small number of patients (level of evidence 3a on the Oxford quality scale). There were no studies evaluating the response to treatment with hypouricemic drugs using MRI. It represents one of the main limitations of this review, which should be kept in mind when evaluating the findings.
Regarding patients studied,9–11 all had gouty arthritis diagnosed by detecting MSU crystals in the synovial fluid with a polarized light microscope, all had baseline uricemia values above EULAR recommendations (>6mg/dL) and in the 2 studies analyzed10,11 all patients had a baseline ultrasound “double contour” sign (deposits of uric acid crystals in hyaline cartilage, which increases the cartilage surface interface thickness reaching something similar to the subchondral bone). Determinations of uric acid at baseline and at each follow-up visit were performed, and hypouricemic treatment was adjusted based on the EULAR recommendations.5,6
Although the included studies were heterogeneous in terms of the clinical and sonographic variables collected, all of them showed the reduction of serum uric acid to be significantly correlated with improved ultrasound findings: decline and even disappearance of the “double contour” sign in patients with uric acid below 6mg/dL in the studies by Thiele and Schlesinger10 and Peiteado et al.,11 and reduction of the maximum transverse diameter and volume of tophi in the study by Perez-Ruiz et al.9
Ultrasound has been shown to be an imaging technique sensitive to change for inflammatory activity present in patients with different inflammatory rheumatic diseases, including gout, allowing us to objectify synovitis or tendinitis and quantify the Doppler signal intensity. It has also proven to be more sensitive than plain X-rays in the early detection of bone erosions, displaying the structural damage to tendons and entheses and being capable of detecting tophi < cm 1, which are difficult to detect even by MRI.12–16
Although based on what has been commented previously it seems logical that ultrasound could be a useful technique in the evaluation of response to treatment with hypouricemic agents in patients with gout, there is little information published in the literature to analyze this aspect. With the studies currently available, we can only say that ultrasound may be useful for evaluating the response to hypouricemic treatment in patients with gout with a 3a level of evidence and a B degree of recommendation. However, taking into account the limitations encountered in this systematic review, we believe that larger studies (randomized clinical trials, cohorts, etc.) will be needed to confirm these results.
As for MRI, although published studies in the literature have shown its reliability in the detection and measurement of tophi, there is currently no evidence to evaluate its usefulness in the treatment response of patients with gout.
Ethical ResponsibilitiesProtection of persons and animalsThe authors state that no experiments were performed on persons or animals for this study.
Data confidentialityThe authors state that they have followed their workplace protocols regarding the publication of patient data and all patients included in the study have received enough information and have given their written informed consent to participate in the study.
Right to privacy and informed consentThe authors state that they have obtained informed consent from patients and/or subjects referred to in this article. This document is in the possession of the corresponding author.
Conflict of InterestThe authors declare no conflicts of interest.
Please cite this article as: Villaverde V, Rosario MP, Loza E, Pérez F. Revisión sistemática sobre el valor de la ecografía y la resonancia magnética nuclear musculoesqueléticas en la evaluación de la respuesta al tratamiento en la gota. Reumatol Clin. 2014;10:160–163.