Clinically evident interstitial lung disease (ILD) affects 10%–42% of RA patients with prognostic implications. The aim of this study was to discern which factors are associated with the presence of ILD in RA patients and to develop a score that could help to stratify the risk of having ILD in RA patients.
MethodsCase–control study. We included RA patients recruited from ILD and rheumatology clinics. We retrieved the following data: gender, age, presence of extra articular manifestations, disease activity scores, antibodies status, ESR, and medication use. Multivariate logistic regression was performed. A risk indicator score was developed.
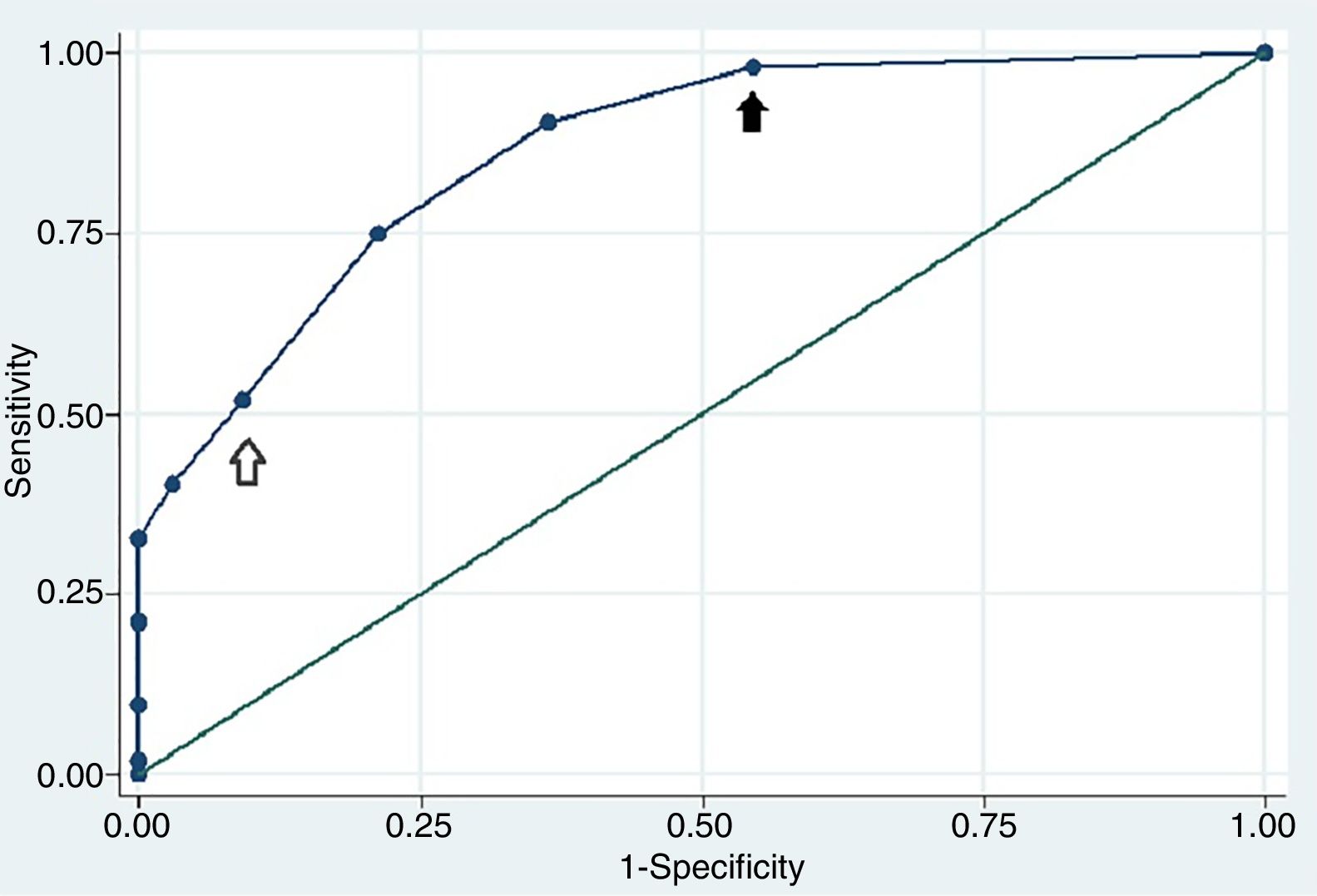
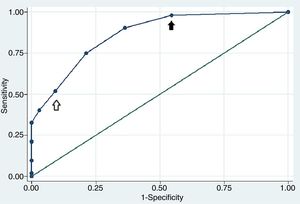
ResultsOf 118 patients included in this study, 52 (44%) had RA-ILD (cases) and 66 (56%) had RA without ILD (controls). Twenty-six patients were male (22%), the mean age was 56.6±15.6 years. Five variables were significantly associated with the presence of ILD: male gender, smoking, extraarticular manifestations, a CDAI score>28, and ESR>80mm/h. The AUC of the final model curve was 0.86 (95%CI 0.79–0.92). Two potential cut-off points of the risk indicator score were chosen: a value of 2 points showed a sensitivity of 90.38% and a specificity of 63.64%, while a value of 4 points showed a sensitivity of 51.9% and a specificity of 90.9%.
ConclusionThis study identified risk factors that could help identify which RA patients are at risk of having ILD through the development of a risk indicator score. This score needs to be validated in an independent cohort.
La enfermedad pulmonar intersticial (EPI) clínicamente evidente afecta al 10-42% de los pacientes con Artritis Reumatoidea (AR), con impacto en el pronóstico. El objetivo de este estudio fue identificar factores asociados y desarrollar una regla para estratificar el riesgo de EPI en pacientes con AR.
MétodosEstudio de casos y controles. Se incluyeron pacientes con AR de una clínica de enfermedades intersticiales y un servicio de reumatología. Se consignaron datos demográficos, manifestaciones extraarticulares, scores de actividad de la enfermedad, autoanticuerpos, tratamiento. Se analizó con regresión logística multivariada.
ResultadosSe incluyeron 118 pacientes con AR, 52 (44%) con EPI (casos) y 66 (56%) sin EPI (controles). Veintiséis (22%) hombres, media de edad 56±15.6 años. Cinco variables se asociaron significativamente con la presencia de EPI: género masculino, tabaquismo, manifestaciones extraarticulares, CDAI> 28, y eritrosedimentación> 80mm/h. El área bajo la curva del modelo final fue 0.86 (IC 95% 0.79-0.92). Se escogieron dos potenciales puntos de corte del score: 2 puntos con una sensibilidad del 90.38% y una especificidad del 63.64%, y 4 puntos con una sensibilidad del 51.9% y una especificidad del 90.9%.
ConclusiónNuestro estudio identificó variables que podrían ayudar a identificar que pacientes con AR se encuentran en riesgo de presentar EPI.
Rheumatoid arthritis (RA) is the most frequent autoimmune disease worldwide, with a prevalence ranging from 0.5 to 1%.1 Airway, lung interstitium and vessels can be affected by the disease, with different prevalence and severity.2 Interstitial lung disease (ILD) is the most severe of these manifestations, with great impact on overall survival and quality of life.3,4 Clinically evident ILD affects 10%–42% of RA patients.5–7 Patients with ILD have three times the risk of death compared with those RA patients without ILD, and ILD is responsible for 7% of all RA associated deaths.6 Given these prognostic implications, it would be useful to identify which patients are at risk of having ILD to facilitate earlier diagnosis and treatment.
Previous studies have shown clinical, demographic and serologic factors are associated with the presence of ILD, including increased age, male gender, smoking, higher disease activity scores, higher values of health assessment questionnaire (HAQ) score, increased erythro sedimentation rate (ESR), positive rheumatoid factor (RF) and antibodies against cyclic citrullinated peptide (CCP). However, available information is not conclusive, and contradictory results are common.8–12 Clinical prediction rules are a useful resource in order to know which patients are at risk of having a specific outcome. The knowledge of isolated risk factors is often not sufficient to determinate the probability of having a specific event in an individual patient. In RA patients with ILD, although prediction rules to identify patients with a progressive fibrosis phenotype have been published,13 to our knowledge, a risk score to identify which RA patients have ILD is lacking. This study aimed to discern which factors are associated with the presence of ILD in RA patients and to develop a score that could help to stratify the risk of having ILD in RA patients.
MethodsThis was a case control study. Cases (RA-ILD patients) were recruited in the ILD clinic of a hospital, in Buenos Aires city, while controls (RA patients without ILD) were recruited in the Rheumatology Division of another hospital, in Salta city during the period between May 2017 and May 2018. The diagnosis of RA was established by ACR 2010 criteria. The diagnosis of ILD in the case patients was defined by HRCT, evaluated by an experienced radiologist. The presence of ILD was excluded in the control patients by directed anamnesis, physical examination, and chest x-ray performed as part of the study. The following data were retrieved from the medical record: gender, age, duration of RA, presence of extra articular manifestations, disease activity scores used in RA (CDAI and DAS28), RF and anti-CCP antibody status, ESR, and medications. The articular evaluation was performed by two independent rheumatologists. It included the number of swelling joints, number of tender joints, and a visual analog scale of the activity of the articular disease. Auto-antibodies determination was made by ELISA CCP2 and immunoturbidimetry for anti-CCP and RF respectively. Extra articular manifestations evaluated were dry eye, dry mouth, and rheumatoid nodules. We recorded medications received for more than 1 months at moment of the evaluation. Smoking status was analyzed including former and past smokers in the “ever smokers” category.
Comparisons between groups were performed using a T-test, Mann–Whitney test, and chi square. Univariate logistic regression was performed, with the presence of ILD as dependent variable. Other variables were analyzed in as independent variables. Continuous variables were categorized according to cut-off points defined by receiver operating curve. Statistical significance was analyzed with Wald test. Variables with P<.1 were considered candidates for the multivariate analysis. The decision to include a variable in the definitive model was guided with the likelihood ratio test. Clinical significance was defined with a P<.05. The Hosmer–Lemeshow goodness of fit test was used to evaluate the model calibration. A risk indicator score was developed, and values were assigned to the included variables according to regression coefficients and their corresponding odd ratios. The discrimination accuracy of the score was expressed as area under the receiver operating curve (AUC). A minimum of 50 cases were included in order to have enough power to build a score with 5 risk factors according to the “one variable per ten events” rule.14
The manuscript was written according to the STROBE initiative for the communication of observational studies.15 The Institutional Ethics Committee of our hospitals reviewed and approved the study.
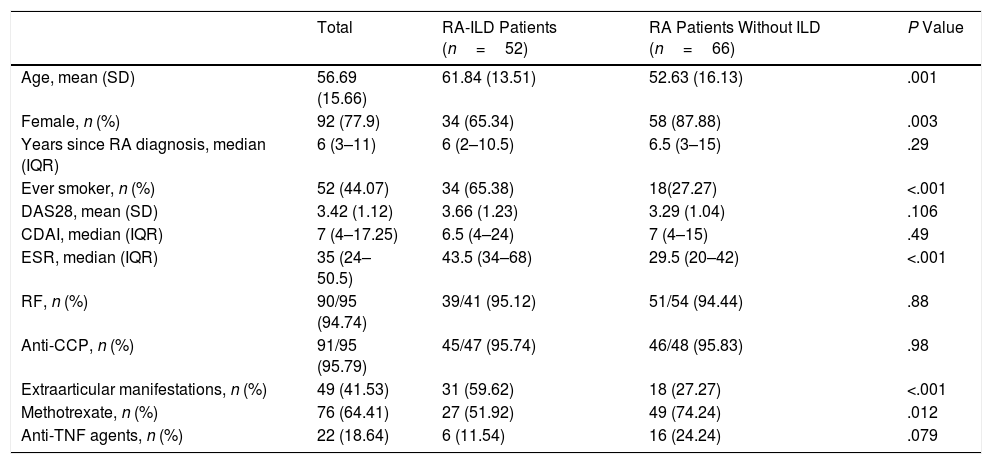
ResultsIn the period between May 2017 and May 2018, 118 patients with RA were seen in the ILD and rheumatology clinics. These were divided into 52 cases (44%) and 66 controls (56%). Baseline characteristics are presented in Table 1. Twenty-six patients were men (22%) and 92 were female (78%). The mean age was 56.6±15.6 years with a median duration of RA of 6 years (IQR 3–11). Ninety-three patients had positive results for anti-CCP (97.8%) and 92 for RF (97.8%).
Demographic and Clinical Characteristics of RA Patients With and Without ILD.
| Total | RA-ILD Patients (n=52) | RA Patients Without ILD (n=66) | P Value | |
|---|---|---|---|---|
| Age, mean (SD) | 56.69 (15.66) | 61.84 (13.51) | 52.63 (16.13) | .001 |
| Female, n (%) | 92 (77.9) | 34 (65.34) | 58 (87.88) | .003 |
| Years since RA diagnosis, median (IQR) | 6 (3–11) | 6 (2–10.5) | 6.5 (3–15) | .29 |
| Ever smoker, n (%) | 52 (44.07) | 34 (65.38) | 18(27.27) | <.001 |
| DAS28, mean (SD) | 3.42 (1.12) | 3.66 (1.23) | 3.29 (1.04) | .106 |
| CDAI, median (IQR) | 7 (4–17.25) | 6.5 (4–24) | 7 (4–15) | .49 |
| ESR, median (IQR) | 35 (24–50.5) | 43.5 (34–68) | 29.5 (20–42) | <.001 |
| RF, n (%) | 90/95 (94.74) | 39/41 (95.12) | 51/54 (94.44) | .88 |
| Anti-CCP, n (%) | 91/95 (95.79) | 45/47 (95.74) | 46/48 (95.83) | .98 |
| Extraarticular manifestations, n (%) | 49 (41.53) | 31 (59.62) | 18 (27.27) | <.001 |
| Methotrexate, n (%) | 76 (64.41) | 27 (51.92) | 49 (74.24) | .012 |
| Anti-TNF agents, n (%) | 22 (18.64) | 6 (11.54) | 16 (24.24) | .079 |
Regarding the HRCT pattern of patients with ILD, 14 patients (26.92%) had a typical usual interstitial pneumonia (UIP) pattern, 18 (34.62%) a possible UIP pattern, and 20 (38.46%) a pattern inconsistent with UIP. With respect to the lung functional tests of patients with ILD, the mean (SD) of FVC% was 71.96 (19.48), FEV1/FVC relation 84.6 (8.35), TLC% 77.27 (15.21), and DLCO% 54.77 (23.28).
Patients with ILD were more likely to be older, male and ever smokers. Also, they had higher values of ESR, and more extraarticular manifestations. Patients without ILD were more likely to have received methotrexate and anti-TNF agents. Three patients received rituximab (1 with ILD), 7 received abatacept (1 with ILD) and 10 received tocilizumab (3 with ILD). With respect to the dose of steroids, patients with ILD were receiving higher doses of prednisone than patients without ILD [mean (SD) 9.76 (1) vs 5.19 (0.93); P<.001). We found no difference between groups in years since RA diagnosis, disease activity scores and anti-CCP and RF status. The articular evaluation was performed by two independent rheumatologists, with an interobserver correlation coefficient (ICC) of 0.84 (CI95% 0.81–0.87).
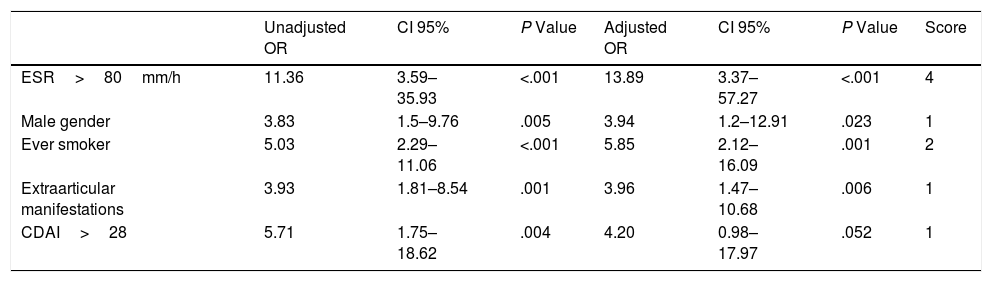
Six variables were associated with the presence of ILD in univariate analysis (Table 2): age>65 years, male gender, ever smoking, extraarticular manifestations (sicca syndrome and/or rheumatoid nodules), a CDAI score>28, and ESR>80mm/h. All these significant variables were included in the initial multivariate analysis, but the variable age was removed in the final model as it lost statistical significance. Although CDAI did not have a significant P-value in the final model, it was included because the likelihood ratio test showed that its exclusion implied a loss of discriminative power to the final model.
Univariate and Multivariate Logistic Regression Analysis With the Presence of ILD as Dependent Variable in Patients With RA.
| Unadjusted OR | CI 95% | P Value | Adjusted OR | CI 95% | P Value | Score | |
|---|---|---|---|---|---|---|---|
| ESR>80mm/h | 11.36 | 3.59–35.93 | <.001 | 13.89 | 3.37–57.27 | <.001 | 4 |
| Male gender | 3.83 | 1.5–9.76 | .005 | 3.94 | 1.2–12.91 | .023 | 1 |
| Ever smoker | 5.03 | 2.29–11.06 | <.001 | 5.85 | 2.12–16.09 | .001 | 2 |
| Extraarticular manifestations | 3.93 | 1.81–8.54 | .001 | 3.96 | 1.47–10.68 | .006 | 1 |
| CDAI>28 | 5.71 | 1.75–18.62 | .004 | 4.20 | 0.98–17.97 | .052 | 1 |
Development of the score. Univariate and multivariate logistic regression analysis showing unadjusted and adjusted OR with their corresponding P value. Values were assigned to the included variables according to regression coefficients and their corresponding odd ratios.
The final clinical model included 5 variables with an AUC of 0.86 (CI 95% 0.79–0.92). In Fig. 1 we show the values assigned to the included variables according to regression coefficients and their corresponding odd ratios. Two potential cut-off points of the score were chosen based on the optimal sensitivity and specificity. A value of 2 points showed a sensitivity of 90.38% and a specificity of 63.64%, while a value of 4 points showed a sensitivity of 51.9% and a specificity of 90.9%. The calibration accuracy of the final model showed good results (Hosmer–Lemeshow goodness of fit test P=.84). An assessment of internal validation with Bootstrap method demonstrated that after 1000 replications, the test demonstrated an AUC of 0.86 (CI 95% 0.80–0.92; P<.001).
Receiver operating characteristic curve of the RA ILD risk indicator score. The area under the receiver operating characteristic curve was 0.86 (CI 95% 0.79–0.92) showing a good discriminatory capacity. A value of 2 points showed a sensitivity of 90.38% and a specificity of 63.64% (black arrow), while a value of 4 points showed a sensitivity of 51.9% and a specificity of 90.9% (white arrow).
This study suggested that certain clinical and demographic factors, including older age, male gender, ever smoking, extraarticular manifestations of RA, elevated CDAI score, and elevated ESR, are associated with the presence of ILD in patients with RA. A risk indicator score based on these risk factors had an AUC of 0.86, demonstrating a good discriminatory capacity to help stratify the risk of having ILD in RA patients. To our knowledge, this is the first risk model developed to help identify the presence of ILD in an RA population and may help clinicians discern which patients could benefit from additional pulmonary screening, such as a high resolution chest CT scan or pulmonary function testing.
Demographic factors associated with the presence of ILD in this RA population are similar to those reported by other authors. Koduri and coworkers carried out a cohort study that showed association between ILD and older age.16 That study showed an increase of 64% of the likelihood of having ILD for each 10 years increase in age. In the study conducted by Kelly and colleagues, male gender and age at RA onset were associated with the presence of ILD.9 Mori and coworkers reported older age as a potent risk factor for ILD with a relative risk ratio of 4.6.17 The association of male gender and ILD has been reported in several studies; however in some of them it did not reach statistical significance.5,16–18 The relationship between smoking and the presence of ILD has also been described by different authors.5,9,19 A group of researchers also found association between subclinical ILD and smoking in RA patients.20 The smoking status not only enhances the risk of having an ILD, but also has impact on functional tests.21 Our study confirms the association between having ILD and these demographic factors (older age, male gender and smoking). However, in the multivariate analysis the variable age lost statistical significance and was removed from the final model.
Controversial results have been reported between the duration of RA and the presence of ILD. Kelly and colleagues reported a median duration of RA at the time of ILD diagnosis of 9 years. In our study, as well as in the one conducted by Koduri, that period was shorter (6 and 3 years respectively). A study that analyzed extraarticular manifestations in RA showed that they are more common in patients with a longer disease duration.22 This contrasts with our results, since we found no difference in the time since the diagnosis of RA between patients with and without ILD.
Factors related to RA activity and/or severity have been reported by different authors as risk factors for ILD. Erosive joint disease, high values of Health Assessment Questionnaire (HAQ), high levels of ESR and presence of rheumatoid nodules have shown association with ILD in two studies.16,23 Disease activity scores (DAS) that evaluate the number of tender and swollen joints together with other activity indicators have shown association with ILD in some but not in all studies.9,16,23,24 Our data shows that patients with high values of CDAI and ESR are at risk of having ILD.
The association between auto-antibodies status (in particular RF and anti-CCP) and ILD is controversial. Although Koduri and coworkers found no association between them, a study conducted by Kelly and colleagues found that patients with ILD were more frequently positive for RF and anti-CCP. Recently, in a case control study with more than 600 patients in each group, the auto-antibodies status sowed no association with the presence of ILD after adjusting for confounding variables.8 The results of this study should be taken into account, considering that it was a multicentric study with a significant number of patients. Yin and collaborators found that anti-CCP (but not RF) was associated to ILD in RA patients.25 Our study found no association between anti-CCP or RF and ILD. This fact may be related to the high prevalence of positive antibodies in control patients.
The inclusion of the control population was made in a consecutive manner in order to avoid selection bias. However, the fact that our rheumatology clinic is a derivation center in this region of the country, could have led to the occurrence of derivation bias, which could explain the high prevalence of positive antibodies and the severity of these patients.
To our knowledge, our study represents the first approach to the development of a score that stratifies the risk of having ILD in RA patients. This is relevant because it can help to determinate witch patients should be submitted to screening tests (HRCT, pulmonary function tests, etc.). A value of 2 points of our score showed a sensitivity of 90.38% and a specificity of 63.64%. This value may be the best cut-off point with the porpoise of using the score to define which patients are candidates to a more specific screening test (i.e. HRCT). The lack of scores designed to guide this decision may be related to the relatively low incidence of clinically evident ILD in this population, which makes difficult to carry out a prospective cohort study. There are few situations in which the screening of ILD is well established. Clear guidelines for screening of ILD in scleroderma has had a great impact on patient management and outcomes, given that ILD is one of the leading causes of death in that population.26–28 ILD may have similar implications in RA patients. An earlier diagnosis could help guide treatment, and more important, avoiding the utilization of potentially harmful agents.29,30 As more data becomes available regarding the optimal treatment of different types of RA-ILD, the timely identification of this extraarticular manifestation of RA will become critical. According to these arguments, we consider that our study can be a contribution of value in this field and that future validation studies are necessary.
This study has the following limitations: (1) A case–control study has inherent limitations when determining the risk of having an event. While a prospective cohort would have been ideal, considering the prevalence and incidence of ILD in this population and the fact that it is not clear at what moment in the evolution of RA ILD develops, a prospective cohort study is difficult to conduct. In this scenario, we believe a case–control study is an acceptable initial approach to build a risk indicator score.2 Our definition of the absence of ILD in control patients may not have enough sensitivity as any ILD below the level of detection with a CXR would have been misclassified. As consequence, it is possible that some of the control patients had subtle ILD, but this would bias the results toward the null and should not affect the significance of our results.3 This score, although internally validated, has not been applied to an external cohort, which may limit the generalizability of our results.4 The utilization of ROC curves to define cut-off points for continuous variables allowed us to make a better categorization of quantitative variables, but implies risk of overfitting.31
In summary, we identified novel risk factors associated with the development of RA-ILD and proposed a risk indicator score to identify those at risk for ILD. Future validation studies in external cohorts are necessary.
Source of FundingThe authors declare that this study didn’t receive support from any organization that needs to be declared.
Conflicts of InterestThe authors have no conflict of interest to declare.