To describe the demographic and clinical characteristics, including health-related quality-of-life (HRQL), in patients with psoriatic arthritis (PsA).
Methods287 patients from 18 Spanish centres were assessed. PsA severity was measured using the following criteria: (1) Psoriasis Area and Severity Index (PASI score 0–72, from low to high severity); (2) number of swollen and tender joints; and (3) Health Assessment Questionnaire (HAQ score 0–3 from low to high impairment in daily activities). HRQL assessment was performed using the following criteria: (a) EuroQol-5D (EQ-5D scores 1–3, with a higher score representing a worse HRQL), Visual Analogue Scale (VAS score 0–100, with a higher score representing a better HQRL) and (b) Short Form-36 (SF-36 score 0–100, with a higher score representing a better HRQL).
Results24.7% of patients were treated with infliximab. In the two groups, 55.7% of the patients were male with a mean age of 52.40±12.53 years. The average number of swollen joints was higher in patients not receiving biological therapy than in those receiving treatment (2.98 vs. 1.54). The mean PASI score was 3.73±5.83, and there was no difference between groups.
HAQ scores were higher in patients receiving infliximab than in those not receiving treatment (0.93 vs. 0.70). The mean EQ-5D scores in the two groups indicated a poorer status based on pain and inability to perform usual/daily activities. HRQL measured by VAS score mean was 60.41±20.08, and there was no difference between the groups. The domains in the SF-36 suggesting poorer functioning in the two groups were the physical role (50.76±43.43), physical pain (49.35±25.69) and the overall physical component (37.88±10.87).
ConclusionsPsA is associated with an impaired HRQL characterised by physical pain and poorer functioning in daily activities.
Describir las características clínicas y demográficas, así como la calidad de vida relacionada con la salud (CVRS), en pacientes con artritis psoriásica (APs).
MétodosSe evaluó a 287 pacientes procedentes de 18 centros españoles. La gravedad de la APs se midió mediante: (1) Psoriasis Area and Severity Index (PASI 0-72, de menor a mayor gravedad); (2) número de articulaciones inflamadas y dolorosas, y (3) Cuestionario de Evaluación de Salud (HAQ), cuyas puntuaciones van de 0 a 3, de baja a gran afectación en las actividades diarias. La CVRS se midió mediante: (a) el cuestionario EuroQol-5D (las puntuaciones del EQ-5D van de 1 a 3; a valores más altos, peor CVRS) y la Escala Visual Analógica (EVA, donde las puntuaciones van de 0 a 100; a valores más altos, mejor HQRL), y (b) SF-36, donde las puntuaciones van de 0 a 100; a valores más altos, mejor CVRS.
ResultadosEl 24,7% de los pacientes fueron tratados con infliximab. En los 2 grupos, la edad media fue de 52,40+12,53 años y un 55,7% fueron varones. El número medio de articulaciones inflamadas fue mayor en los pacientes que no recibieron infliximab (2,98 vs. 1,54). La media de las puntuaciones en el PASI fue de 3,73 ± 5,83, sin diferencias entre grupos.
Las puntuaciones en el HAQ fueron mayores en los pacientes con infliximab (0,93 vs. 0,70). Las puntuaciones medias de EQ-5D en los 2 grupos que indican peor estado de salud fueron: el dolor y la incapacidad para realizar actividades diarias. La CVRS medida por la puntuación media de la EVA fue 60,41±20,08, sin diferencias entre grupos. En el SF-36, las dimensiones con valores más bajas en los 2 grupos fueron: función física 50,76±43,43, dolor corporal 49,35±25,69 y el componente físico general 10,87±37,88.
ConclusionesLa APs se asocia a un deterioro de la CVRS caracterizada por dolor corporal y peor funcionamiento en las actividades diarias.
Psoriatic arthritis is a seronegative chronic inflammatory arthritis associated with psoriasis.1 In most patients, there is no direct relationship between psoriasis severity and arthritis.2 The exact prevalence is unclear due to the heterogeneity of this disease in clinical presentation. Available estimates of prevalence range from 0.04 to 0.1%.3 The prevalence of psoriasis varies from 2 to 3% of the population, with 4–42% of psoriatic patients having psoriatic arthritis.3 A study performed in Spain on the prevalence of psoriasis showed that 10% of patients with psoriasis reported arthralgias; furthermore, a diagnosis of psoriatic arthritis was established in 9.4% of these patients, and a joint deformity was present in 4.9% of these patients.4
A better understanding of the demographic and clinical characteristics of psoriatic arthritis patients is important to improve the management of this illness. Several studies have shown that patients with moderate-to-severe psoriatic arthritis have significant impairment in quality-of-life compared to the general population.1,5–11
Patients with psoriatic arthritis have joint impairment and significant physical limitations. Several studies have shown that, in patients with psoriatic arthritis, the impairment in physical activity is greater than the effect on the mental health of the patient.12,13 In psoriatic arthritis, disturbances in joint function are associated with body pain and loss of mobility, which result in impairment in usual daily activities. The disability generated by the illness is, in some cases, associated with difficulties in employment, and patients may have to change their work or abandon their current employment, often with a concomitant decrease in income. Additionally, a higher risk of mortality has been observed in patients with active and severe psoriatic arthritis compared to the general population.14
To provide reliable data about psoriatic arthritis patients, the objectives of the present study were to describe patient characteristics, assess their health-related quality-of-life (HRQL), and assess the effect of infliximab in patients with psoriatic arthritis in Spain.
MethodsStudy designA cross-sectional, observational, multi-centre study was conducted involving 18 centres in Spain. Information on patient clinical characteristics and HRQL was collected by a dermatologist and rheumatologist working together in each centre. The study was approved by the Clinical Research Ethics Committee for all of the participating centres, and informed consent was signed by all of the subjects in the study. The recruitment period was from January to March 2007.
The patient inclusion criteria were the following: (1) age >18 years; (2) a diagnosis of psoriatic arthritis confirmed by a dermatologist and rheumatologist; and (3) capacity to understand the questionnaires used in the study. The exclusion criteria were the following: (1) current participation in a clinical trial; (2) receiving biological therapy apart from infliximab; and (3) axial disease involvement.
Assessment of demographic and clinical characteristicsThe demographic data collected in the study included age, gender, illness duration, weight, date of the psoriatic arthritis diagnosis and loss of productivity.
Three different measures were used to describe the severity of psoriatic arthritis: (1) Psoriasis Area and Severity Index (PASI); (2) Health Assessment Questionnaire (HAQ); and (3) number of swollen and tender joints.
The PASI score considers the severity of psoriasis as a function of the area of skin involved and the grade of erythaema, desquamation and/or infiltration.15 The final score has a range from 0 to 72, which is the weighted sum of different area scores. The affected surface is measured in 4 different areas, head, trunk, upper extremities and lower extremities, each scored between 0 and 6. The erythaema, desquamation and infiltration scores range between 0 and 4.
The HAQ assesses impairment in daily activities due to illness.16 The items contained in the HAQ are a sample of 20 daily activities divided into 8 different dimensions (dressing and grooming, awakening, eating, walking, hygiene, reach, grip and other activities). The patient can choose between 4 different responses (no difficulty, some difficulty, considerable difficulty and incapable). Apart from these questions, the patients are asked some questions regarding the need for physical aids or assistance from another person to perform these activities. Each HAQ dimension score ranges from 0 (without impairment) to 3 (unable to do), and the summary score is the mean of the 8 HAQ dimensions.
A printed picture of the human body was used by the researchers to help the patient to identify and record the number of swollen and tender joints.
Assessment of the health-related quality-of-life (HRQL)Two questionnaires were used to define the HRQL: (1) EuroQol-5D (EQ-5D), and (2) Short-Form 36 (SF-36). The EQ-5D is composed of 5 items, each one assessing different dimensions of quality-of-life in a range of 1 to 3.6,17,18 A score of 3 for an item represents the worst level of function. The final score is descriptive and is represented by five numbers, for example, 11213, with each number representing the score for each item. Utility values can be computed based on these scores.19 The utility value represents the patient's health state between 0 (death) and 1 (best possible health state). Furthermore, a Visual Analogue Scale (VAS) was included in the questionnaire. The patients used this scale to self-assess global HRQL. The scale is represented by a printed thermometer with a scale ranging from 0 (poorest quality-of-life) to 100 (best possible state of quality-of-life).
In the SF-36, the patients graded 36 items representing 8 different HRQL domains.5,8,9,20,21 The scores were transformed onto a 0 to 100 scale, with higher scores representing the best self-assessment for the domain. Two summary factors were computed using this questionnaire: physical component summary (PCS) and mental component summary (MCS). These components were generated by a factor analysis that has been validated in Spain.8,22
Data analysisStatistical analyses were performed using PASW® 18.0 (Predictive Analytics Software) software. Descriptive statistics and statistical tests were performed. Categorical variables were assessed using the chi squared test or Fisher exact test, and quantitative variables were analysed using a Student t test or ANOVA. The level of significance was set to 0.05.
To estimate the influence of demographic and clinical variables on the impairment of daily activities (measured by HAQ) and HRQL (measured by EQ-5D and SF-36), multivariate analysis (multivariate linear regressions) was performed. The specified dependent variables were summary HAQ score, EQ-5D utility values, and the PCS and MCS from the SF-36 questionnaire.
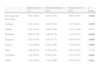
ResultsCharacteristics of the patients with psoriatic arthritisThe study included 287 patients with psoriatic arthritis, with 71 (24.7%) patients receiving infliximab. The patient demographics are shown in Table 1. The mean (±SD) age was 52.4 (±12.5) years, and the mean weight was 76.5 (±15.1)kg. Additionally, 56% of the patients were male. Of the 287 patients, 16 (6%) lived alone. No significant differences were found in the demographic characteristics between the patients receiving or not receiving infliximab.
Demographic and clinical characteristics (n=287).
| Infliximab (n=71) | Non-infliximab (n=216) | Total score (n=287) | p-Value | |
| Age, years | ||||
| Mean (SD) | 51.54 (12.71) | 52.61 (12.48) | 52.40 (12.53) | 0.510 |
| Weight, kg | ||||
| Mean (SD) | 76.64 (1.69) | 76.42 (15.45) | 76.47 (15.14) | 0.915 |
| Males, % | 55.6 | 56.3 | 55.7 | 0.510 |
| Disease duration, years | ||||
| Mean (SD) | 12.56 (11.51) | 10.29 (8.88) | 10.85 (9.62) | 0.087 |
| Number of swollen joints | ||||
| Mean (SD) | 1.54 (2.57) | 2.98 (4.38) | 2.62 (4.05) | 0.009 |
| Number of tender joints | ||||
| Mean (SD) | 4.00 (5.82) | 5.28 (7.14) | 4.97 (6.85) | 0.172 |
| PASI | ||||
| Mean (SD) | 2.63 (5.00) | 4.09 (6.04) | 3.73 (5.83) | 0.067 |
| Patients with PASI<10, % | 93% | 90.70% | 91.3% | 0.582 |
| Patients with 10≤PASI≤20,% | 4.20% | 7.40% | 6.6% | |
| Patients with PASI>20, % | 2.80% | 1.90% | 2.1% | |
| Patients treated with infliximab (%) | 100% | 0% | 24.7% | |
Bolded p-values represent statistical significance.
No significant differences were found between the patients receiving or not receiving infliximab for clinical variables, except for the number of swollen joints. The patients receiving infliximab had a lower mean number of swollen joints than patients not receiving infliximab (1.54 vs. 2.98) (Table 1). The mean (±SD) years of disease duration was 10.9 (±9.6). The mean number of tender joints was 5.0 (±6.9), with a maximum number of 49 tender joints.
The mean (±SD) PASI score was 3.7 (±5.8), with a minimum score of 0 and a maximum score of 51. The mean PASI cut-off score for moderate severity psoriatic symptoms was <10. The majority of patients (91.3%) had a PASI score below 10, 6.6% of patients had a PASI score of 10–20 and only 2.1% of patients had a PASI score greater than 20.
Impairment in daily activities and work due to psoriatic arthritisThirty percent of patients reported that, on some days during the previous month, they were unable to perform standard daily activities. The mean number (±SD) of days per month that patients were unable to do their daily activities was 12.8 (±10.7), and the hours per day that patients felt unable to do their daily activities was 9.0 (±7.1). Additionally, 21% of patients felt the need to request help from friends/family to perform the most common daily life activities.
Of the 287 patients, 55% were employed at the time of the study, and 16% retired early due to the psoriatic arthritis (Table 2). Six percent of patients had changed their job because of the illness. The mean (±SD) reduction in working hours per week was 0.8 (±3.3), and the mean (±SD) income reduction per month was 7.6 (±52.9) euros/patient. No significant differences were found with regard to productivity loss between the patients receiving or not receiving infliximab.
Work impairment characteristics (n=287).
| Infliximab (n=71) | Non-infliximab (n=216) | Total score (n=287) | p-Value | |
| Work status, % | 0.671 | |||
| Employed | 50.7 | 56.5 | 55.1 | |
| Unemployed | 2.8 | 0.9 | 1.4 | |
| Housewife | 14.1 | 16.2 | 15.7 | |
| Disabled | 26.8 | 12 | 15.7 | |
| Retirement | 5.6 | 14.4 | 12.2 | |
| Days per month without working | ||||
| Mean (SD) | 1.80 (6.51) | 0.82 (3.92) | 1.06 (4.70) | 0.127 |
| Range | 0–30 | 0–30 | 0–30 | |
| Hours per week without working | ||||
| Mean (SD) | 0.53 (2.14) | 0.83 (3.54) | 0.76 (3.26) | 0.504 |
| Range | 0–12 | 0–30 | 0–30 | |
| Income reduction, € per month | ||||
| Mean (SD) | 8.45 (49.99) | 12.36 (75.08) | 7.56 (52.84) | 0.682 |
| Range | 0–600 | 0–600 | 0–600 | |
| Need help from relatives or friends,% | 31 | 18.1 | 21.3 | 0.061 |
Significant differences were found in almost all of the HAQ dimensions between the patients receiving and not receiving biological therapy. However, patients receiving infliximab had higher impairment in daily activities than patients managed without infliximab (overall mean HAQ 0.93 vs. 0.70, p=0.011). The detailed results are shown in Table 3.
HAQ score.
| Infliximab (n=71) | Non-infliximab (n=216) | Total score (n=287) | p-Value | |
| Dressing and grooming | 0.91 (0.92) | 0.55 (0.79) | 0.64 (0.84) | 0.020 |
| Arising | 0.91 (0.91) | 0.67 (0.84) | 0.73 (0.87) | 0.039 |
| Eating | 0.94 (1.02) | 0.98 (0.99) | 0.97 (1.00) | 0.810 |
| Walking | 0.69 (0.78) | 0.46 (0.75) | 0.52 (0.77) | 0.030 |
| Hygiene | 0.83 (0.89) | 0.52 (0.77) | 0.60 (0.81) | 0.005 |
| Reach | 1.20 (0.90) | 0.85 (0.89) | 0.94 (0.91) | 0.005 |
| Grip | 0.90 (0.94) | 0.79 (0.94) | 0.81 (0.94) | 0.370 |
| Activities | 1.08 (0.91) | 0.80 (0.84) | 0.87 (0.87) | 0.016 |
| Total | 0.93 (0.69) | 0.70 (0.66) | 0.76 (0.67) | 0.011 |
Bolded p-values represent statistical significance.
Differences in HRQL measured by the EQ-5D questionnaire were found in the dimensions of self-care and pain/discomfort. Patients with infliximab scored higher on the self-care dimension, and patients with non-biological therapy scored higher in the dimension of pain/discomfort.
The dimensions most affected in the two groups of patients with psoriatic arthritis (as measured by EQ-5D) were pain and daily activities. In our sample, 68% of the patients receiving infliximab and 79% of those not receiving infliximab suffered from moderate or severe pain. The pain dimension was followed by daily activities (a score of 1.5±0.6) as the most affected dimension. There were no significant differences between the two groups. Thus, 48% of patients reported that they had some difficulties or that they felt unable to perform their daily activities. The mean (±SD) utility of the patients with psoriatic arthritis was 0.67 (±0.23). The patients’ global health state, as measured by the VAS, showed a mean (±SD) score of 60.4 (±20.1). Detailed descriptive statistics of the EQ-5D are summarised in Table 4.
EQ-5D questionnaire (n=287).
| EQ-5D | Infliximab (n=71) | Non-infliximab (n=216) | Total score (n=287) | p-Value |
| Mobility | ||||
| Mean (SD) | 1.57 (0.50) | 1.44 (0.53) | 1.48 (0.52) | 0.066 |
| Self-Care | ||||
| Mean (SD) | 1.43 (0.53) | 1.26 (0.46) | 1.30 (0.48) | 0.014 |
| Usual activities | ||||
| Mean (SD) | 1.61 (0.55) | 1.47 (0.55) | 1.52 (0.55) | 0.087 |
| Pain/discomfort | ||||
| Mean (SD) | 1.74 (0.58) | 1.90 (0.55) | 1.86 (0.56) | 0.043 |
| Anxiety/Depression | ||||
| Mean (SD) | 1.56 (0.65) | 1.43 (0.58) | 1.48 (0.60) | 0.106 |
| Utility | ||||
| Mean (SD) | 0.64 (0.25) | 0.68 (0.23) | 0.67 (0.23) | 0.215 |
| VAS | ||||
| Mean (SD) | 60.46 (19.79) | 60.39 (20.22) | 60.41 (20.08) | 0.978 |
Bolded p-values represent statistical significance.
The results showed that the most affected dimensions were role-physical, physical pain, general health, and vitality. The role-physical evaluates how physical health affects the performance of work and other daily activities. The mean (±SD) score for the role-physical was 50.8 (±43.4). Physical pain is a measure of pain severity and its effects on working, including domestic labour. The mean (±SD) value for this dimension was 49.4 (±25.7). The worst score [mean (±SD)] was found for the general health dimension [43.8 (±19.8)], which represents the patient's self-assessment of current health, future health options, and resistance to illness. Vitality, which is a dimension that assesses the feeling of well-being and energy, had a mean (±SD) score of 50.0 (±22.6). No significant differences were found between the two groups of patients.
All of the dimensions in the SF-36 questionnaire can be summarised into 2 different, independent factors: the PCS and MCS. The mean (±SD) values were 37.9 (±10.9) and 46.9 (±12.6) for the PCS and MCS, respectively. These results show a greater impairment in the PCS than the MCS. The descriptive statistics from the SF-36 are shown in Table 5.
SF-36 questionnaire (n=287).
| SF-36 | Infliximab (n=71) | Non-infliximab (n=216) | Total score (n=287) | p-Value |
| Physical functioning | ||||
| Mean (SD) | 56.27 (28.10) | 63.16 (26.35) | 61.64 (26.84) | 0.061 |
| Role-physical | ||||
| Mean (SD) | 46.83 (43.70) | 52.05 (43.70) | 50.76 (43.43) | 0.384 |
| Physical pain | ||||
| Mean (SD) | 50.34 (26.60) | 49.02 (25.43) | 49.35 (25.69) | 0.709 |
| General health | ||||
| Mean (SD) | 41.91 (19.40) | 43.99 (20.35) | 43.77 (19.75) | 0.452 |
| Vitality | ||||
| Mean (SD) | 48.43 (24.53) | 50.30 (22.06) | 50.01 (22.58) | 0.549 |
| Social functioning | ||||
| Mean (SD) | 67.86 (28.12) | 72.91 (27.95) | 72.81 (27.53) | 0.191 |
| Role-emotional | ||||
| Mean (SD) | 66.67 (43.64) | 74.37 (39.86) | 72.91 (40.83) | 0.170 |
| Mental health | ||||
| Mean (SD) | 61.49 (21.62) | 64.35 (20.66) | 63.99 (20.67) | 0.320 |
| Physical component summary (PCS) | ||||
| Mean (SD) | 36.88 (11.18) | 38.18 (10.76) | 37.88 (10.87) | 0.385 |
| Mental component summary (MCS) | ||||
| Mean (SD) | 44.999 (12.80) | 47.29 (12.60) | 46.90 (12.59) | 0.189 |
The multivariate analysis showed that the main factors that affected the performance of the daily activities measured by the HAQ were receiving infliximab, sex, age and the number of swollen and tender joints. From these variables, the major influence was due to the sex and receiving biological therapy. Men receiving infliximab had greater impairment in daily activities.
Regression analyses with HRQL as a dependent variable showed that patients with a higher number of tender joints had a worse HRQL. Pain was the most influential variable on HRQL measured using the EQ-5D and SF-36. Other variables with some influence in HRQL were sex and age. Older patients and women have a higher risk of HRQL impairment. Detailed results of multivariate analyses are shown in Table 6.
Multivariate analysis (n=287).
| Beta coefficients | p-Value | Adjusted R squared | |
| HAQ model | |||
| Infliximab | 0.312 | <0.001 | 0.193 |
| Weight | 0.004 | 0.137 | |
| Age | 0.011 | <0.001 | |
| Sex | −0.372 | <0.001 | |
| Swollen joints | 0.020 | 0.047 | |
| Tender joints | 0.023 | <0.001 | |
| PASI | 0.004 | 0.490 | |
| EQ5D model | |||
| Infliximab | −0.122 | 0.031 | 0.137 |
| Weight | −0.069 | 0.288 | |
| Age | −0.058 | 0.295 | |
| Sex | 0.152 | 0.020 | |
| Swollen joints | −0.062 | 0.310 | |
| Tender joints | −0.327 | <0.001 | |
| PASI | −0.066 | 0.244 | |
| SF36 model (physical component) | |||
| Infliximab | −0.096 | 0.087 | 0.163 |
| Weight | −0.052 | 0.424 | |
| Age | −0.127 | 0.023 | |
| Sex | 0.216 | 0.001 | |
| Swollen joints | −0.044 | 0.470 | |
| Tender joints | −0.325 | <0.001 | |
| PASI | −0.099 | 0.078 | |
| SF36 model (mental component) | |||
| Infliximab | −0.083 | 0.174 | 0.014 |
| Weight | 0.020 | 0.778 | |
| Age | 0.074 | 0.219 | |
| Sex | 0.016 | 0.824 | |
| Swollen joints | 0.019 | 0.774 | |
| Tender joints | −0.173 | 0.008 | |
| PASI | 0.008 | 0.901 | |
Bolded p-values represent statistical significance. Infliximab was coded as 0 patients not receiving infliximab and 1 patients receiving infliximab. Sex was coded as 0 female and 1 male.
To our knowledge, our study is the first one performed in Spain to measure HRQL only in psoriatic arthritis. Other publications23,24 included a mixed psoriasis and psoriatic arthritis population and did not categorise the results by disease.
The most important impairment observed in the study was related to work and daily activities. The main problem communicated by the patients was the impairment that arthropathy causes on normal daily living. Patients sometimes feel unable to perform some activities, such as vacuuming, rising from bed, or getting in and out of a car. The difficulties associated with these activities are attributable to joint movement and/or resistance. Thus, activities that force joint resistance are the most difficult for these patients to perform.
In patients with psoriatic arthritis, pain has a high impact on usual daily activities.2 The effect of tender joints is reflected in the EQ-5D questionnaire, in which the mean value for the pain item is the worst reported. Additionally, the physical pain dimension had one of the worst values in the SF-36 questionnaire. Multivariate analysis confirmed this tendency; the variable with the greatest influence in the HRQL models was the number of tender joints. Therefore, we can conclude that pain is a key factor in the impairment of quality-of-life in these patients.
General health was highly impaired in patients with psoriatic arthritis, as assessed by the VAS (EQ-5D questionnaire) and SF-36. The worst mean value (43.8) was reported for the dimension of general health in the SF-36. The mean VAS value (60.4) was similar to that observed for other chronic diseases.17
Impaired physical function leads to a loss of productivity. Some patients are obliged to change their work, reduce their working hours, or sometimes even quit their jobs because of the illness. These problems lead to decreased patient income. Thus, the loss of productivity is another important factor related to the impairment in quality-of-life. These results are also demonstrated in pharmacoeconomic studies.25
Comparisons with other studies are difficult due to differences in the methodology and treatments.26 The mean age at first symptom onset (52.4 years) and the proportion of males (56%) were similar compared to other studies of psoriatic arthritis.13,27,28 These results confirm that psoriatic arthritis occurs with roughly a similar frequency in both sexes.
Due to the dual component of psoriatic arthritis (dermatologic and rheumatologic), disease severity can be established using different parameters. Based on PASI scores, the disease severity in our study sample would be defined as low because most of the patients had a PASI score <10 points. Reich et al.13 analysed a general German sample and reported higher scores (mean 14.3). Another study performed by Brodsky et al.28 also had a higher PASI score than our study (6.5±8.7).
Arthropathy is established by the number of tender and swollen joints. The reported number of tender joints in our sample was 4.97±6.85. This number is relatively less than that reported by other authors (7.7±8.7 joints).28 In our data, the patients receiving infliximab had a better clinical profile than the patients not receiving therapy.
In terms of health-related quality-of-life, the results of the present analysis showed an EQ-5D mean VAS of 60.4 and a mean utility of 0.67. Our patients had a better overall health HRQL measured using EQ-5D compared to the results reported by Wade et al.29 for 44 patients who began treatment with etanercept (EQ 5D mean VAS of 45.6±19.2 and an EQ 5D mean utility of 0.42±0.32).
Adams et al.27 recruited psoriatic arthritis patients from an Irish rheumatology referral centre who began treatment with biological therapy. At baseline, the patients reported an EQ-5D mean utility of 0.49±0.32, which was lower than that in our study. However, the EQ-5D mean utility improved to 0.77±0.28 after twelve months of biological therapy. These differences between the baseline and the follow up could be explained by the introduction of biological therapy during the study period.
Analysis of health-related quality of life measured using SF-36 provided results similar to those reported by Adams et al.27. The SF-36 mean PCS was lower than the MCS, suggesting that the physical disturbance in psoriatic arthritis is the main factor that affects the HRQL. Unfortunately, Adams’ study did not report the follow-up results for the SF-36 components, which could have allowed for comparisons of the results in the treated patients.
The use of biological therapy to treat psoriatic arthritis has changed the management of patients with psoriatic arthritis. The effect of infliximab in the present study was found in the clinical parameters; however, the functional status was worse in these patients. One reason could be because in this study infliximab was only used to manage severe patients. Therefore, these patients had a better clinical status, but infliximab could not recover their daily life functioning.
The main limitation of the present study could arise from the cross-sectional design, which avoids performing comparisons between data over time.
Concomitance of psoriasis and psoriatic arthritis can cause confusion in the collected data. These misdiagnoses can introduce a bias into the results of studies that include patients with psoriasis and psoriatic arthritis; in some patients, the psoriasis diagnosis overlaps with psoriatic arthritis. The number of patients diagnosed with psoriatic arthritis can vary depending of the criteria used. Therefore, using reliable criteria to diagnose psoriatic arthritis is important. An advantage of our study is that we focused only on psoriatic arthritis patients; thus, the risk of a psoriasis diagnosis overlap with psoriatic arthritis was reduced.
Despite the mentioned limitations, our study provides detailed data on the characteristics of psoriatic arthritis patients in Spain that could be interesting for clinicians and other stake holders. The results of the present work show that psoriatic arthritis has a substantial impact on quality-of-life. Furthermore, because of the physical difficulties associated with the disease, these patients suffer a loss in their economic activity and a reduction in income. Physical pain and disability are the most important factors impacting a patient's quality-of-life.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the responsible Clinical Research Ethics Committee and in accordance with those of the World Medical Association and the Helsinki Declaration.
Confidentiality of dataThe authors declare that they have followed the protocols of their work centre on the publication of patient data and that all the patients included in the study have received sufficient information and have given their informed consent in writing to participate in that study.
Right to privacy and informed consentThe authors have obtained the informed consent of the patients and/or subjects mentioned in the article. The author for correspondence is in possession of this document.
FundingThis study received an unconditional grant from Schering-Plough. The authors had independence from the funding body with respect to study design, analysis and interpretation of the data, report writing, and submission of the manuscript for publication.
Conflict of interestThe authors declare no conflict of interest.
The authors thank Eliazar Sabater and Covadonga Torres of Pharmacoeconomics and Outcomes Research Iberia (PORIB) for their assistance with the statistical analysis and manuscript writing.