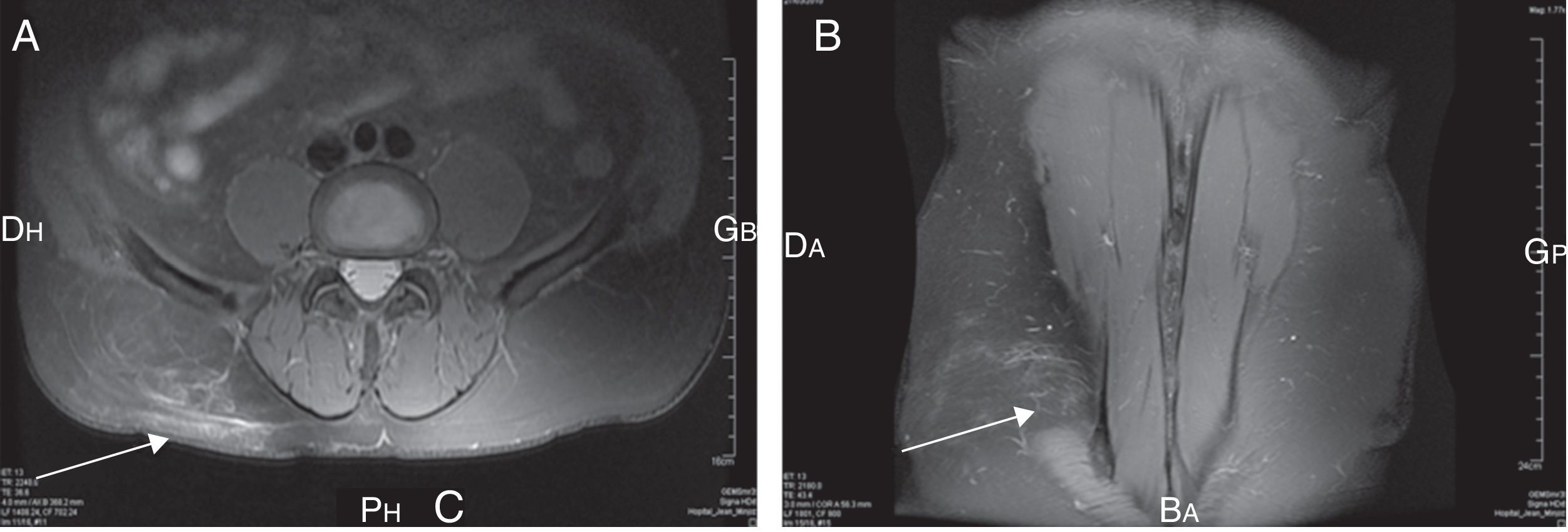
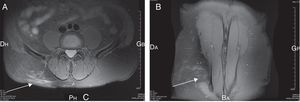
A 67-year-old male patient presented with acute thoraco lumbar and right trochanteric pain at the consultation of Rheumatology. The patient could not sleep due to the pain, mostly localized in the right iliac crest but irradiating to the trochanter. Pain was aggravated by pressure, right side-lying and seating postures. Magnetic resonance imaging of the spine revealed a lumbar myositis, with T2 high signals in the paraspinal muscles (L3–L4 area) (Fig. 1). There was no cutaneous eruption, no neck stiffness, no biologic inflammatory syndrome, no biologic sign of muscular lysis and no hypereosinophilia. Results of immunological investigations including complement fractions explorations, rheumatoid factor dosage and cryoglobulins detection, were negative. Results of serum antibody tests for Borrelia burgdorferi, Coxiella burnetti and Treponema pallidum were also negative. In contrast, the Toxocara canis serologic tests were strongly positive by both Toxocara enzyme-linked Immunosorbent assay and Toxocara Western blot assays. The patient did not have a dog but was living in a rural area and reported frequent contacts with soil. Pains disappeared rapidly after 4 days of albendazole treatment. Six months later, the patients relapsed with similar symptoms in intensity and location; the pains disappeared again after 4 days of albendazole treatment.
Human toxocariasis is a widespread soil-transmitted parasitic disease. Principal risk factors include contact with dogs and soil and residence in a rural region.1,2 After ingestion Toxocara spp. embryonated eggs hatch in the intestines and the larvae migrate via the blood circulation to the liver, lungs and left heart, from where they disseminate via the systemic circulation to eyes, brain or muscles.1,2Toxocara spp. infections are highly pleiomorphic, depending on the number and location of encysted larvae in the body and the immune sensitivity of the host.
Definitive diagnosis of toxocariasis would require identification of larvae in human tissues such as skin biopsies, but this is rarely achieved. Besides, as Toxocara spp. infection is a parasitological dead-end, neither adult roundworms nor eggs can be found in stools to make a direct diagnosis. Consequently, the diagnosis is usually made on the basis of serologic tests results. Two types of tests are available for the immunodiagnosis of toxocariasis: the ELISA and Western blot (WB), both using T. canis excretory-secretory antigens (TES). WB is the more sensitive and specific of the two assays.3,4
Amongst all available drugs, albendazole, is the most commonly used and treatment of choice because its reaches higher serum levels, is well-tolerated and better absorbed outside the gastronintestinal tract.2,5 Repeated courses of anthelmintic treatments may be necessary to eliminate definitely the symptoms.6,7 Pharmacological treatment must be accompanied by prevention of recontamination by deworming dogs and sanitary education.1,6
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this investigation.
Confidentiality of dataThe authors declare that no patient data appears in this article.
Right to privacy and informed consentThe authors declare that no patient data appears in this article.
Conflicts of interestThe authors have no conflicts of interest to declare.