It is quite clear that medicine is biased towards positive results and the same applies to the practice of pathology.1 One of the ubiquitous tests in autoimmunity, the antinuclear antibody (ANA) suffers from this very same fate. A number of guidelines report on the clinical utility of a positive ANA and dissuade clinicians from requesting this test in the setting of low pre-test probability for an ANA-associated autoimmune disorder (AAD).2 This is certainly sound advice and prevents unnecessary investigations and healthcare expenditure. Yet, it is important to realise the clinical importance and pitfalls of a negative ANA results which sometimes becomes forgotten.
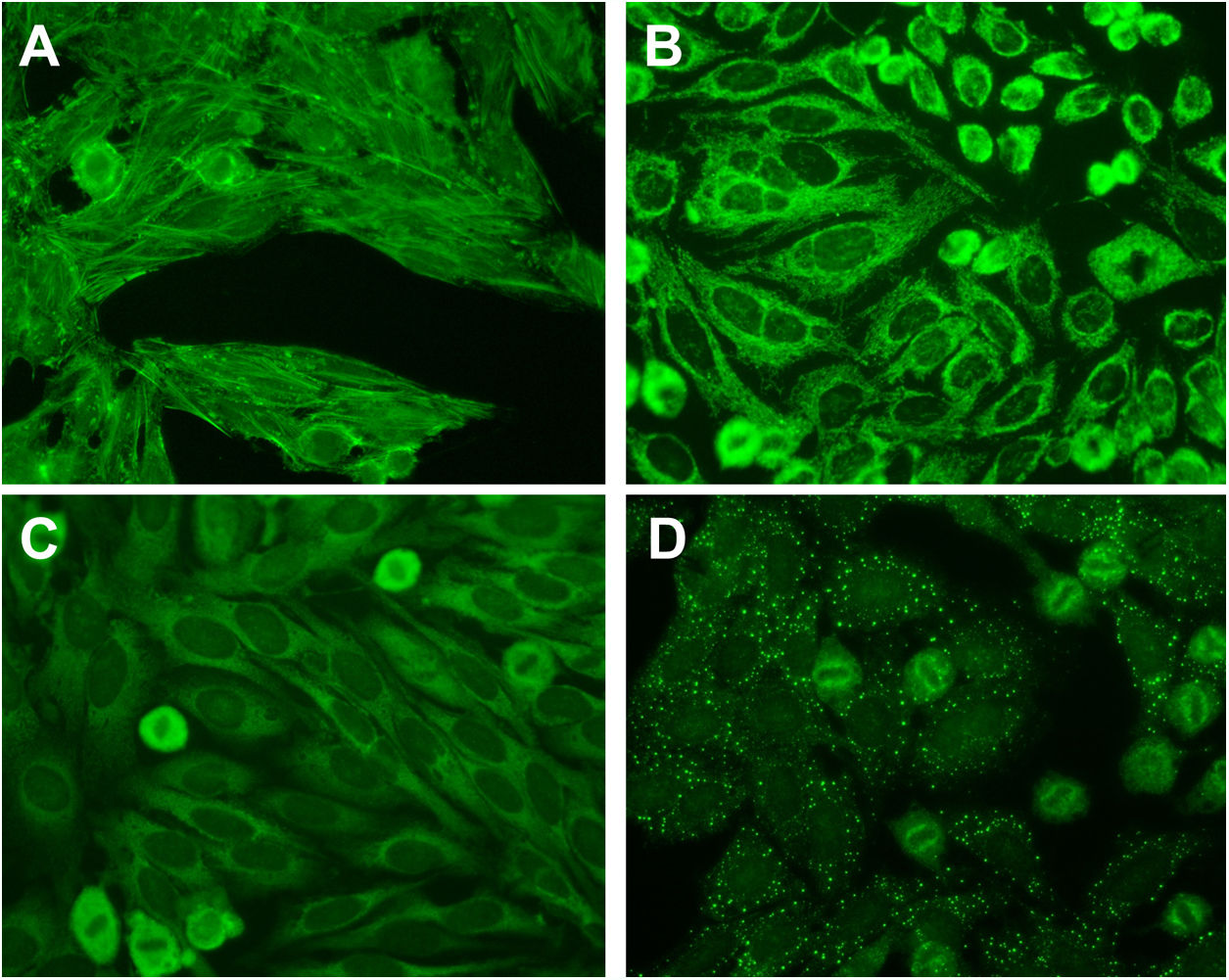
The internationally-accepted “gold standard” to measure ANA is via indirect immunofluorescence on HEp-2 cells.3 A negative ANA test on HEp-2 substrate usually means that there is no significant detection of IgG ANA (in the nucleus) at a specified dilution of serum – usually 1:80 to 1:160. There is a move to also classify positive cytoplasmic and mitotic staining of the HEp-2 substrate as ANA positive.3,4 This may improve the sensitivity of detecting AADs and prompt appropriate further testing and follow-up (Fig. 1).3
Example cytoplasmic staining on the HEp-2 substrate. (A) F-actin staining suggesting the presence of smooth muscle antibodies found in autoimmune hepatitis and related disorders. (B) Coarse, granular cytoplasmic staining suggestive of anti-mitochondrial antibodies found in primary biliary cirrhosis. (C) Smooth, homogenous cytoplasmic staining suggestive of anti-ribosomal P antibodies found in systemic lupus erythematosus. (D) Large cytoplasmic dots staining suggestive of anti-GW bodies. All micrographs are taken at a magnification of 400×.
The high sensitivity and negative predictive value (NPV) for systemic lupus erythematosus (SLE) makes the ANA test a good “rule out” test to essentially exclude this disorder if it is negative.5 Indeed, the most recent European League Against Rheumatism/American College of Rheumatology guidelines for the diagnosis of SLE mandates a positive ANA (>1:80) on the HEp-2 substrate to be considered for this diagnosis.6 Sensitivities for detecting other AADs is low-moderate at best; yet also demonstrates very high NPVs.7 Unless there has been a significant change in clinical picture or there is a suspicion of a laboratory issue, there is little value in repeating an ANA that is initially negative.8
A pitfall is that ANA is a screening test and may, in rare instances, miss low-level specific autoantibodies/anti-extractable nuclear antigens (ENAs) if more sensitive assays are not performed,9 or miss anti-ENA that do not produce a characteristic ANA pattern e.g., anti-Ro52. Therefore, the substrate should be specified in the report since substrates such as the HEp-2000® (Immunoconcepts) which has transfected Ro60 increase the detection of anti-Ro60 and hence, a negative result makes the presence of anti-Ro60 less likely.10
If there is a high clinical suspicion for an AAD, the clinician should request further anti-ENA tests and the overall clinical picture and physician's interpretation of the patient should prevail. This is especially of importance since commercial HEp-2 substrates, whilst generally demonstrating excellent inter-assay and inter-laboratory agreement, display subtle staining differences that affect the microscopist's final interpretation.11 The significance of low levels of anti-ENA with negative ANA is not well established.
To conclude, clinicians should be aware of the value, implications and pitfalls of a negative ANA result when considering AADs. They should also be aware of their laboratory's definitions of a “negative” ANA result, the substrate used and whether they report non-nuclear patterns which may have important implications for their patients. Importantly, the overall clinical picture of the patient should be taken into considerations when deciding on the relevancy of a negative ANA test.
FundingNil.
Conflict of interestsNone declared.