In the ARCO study, adherence to subcutaneous biological agents by patients with rheumatoid arthritis improved with monthly administration. We assess whether adherence can be related to fulfilment of expectations and satisfaction with treatment.
Patients and methodsAdherence was assessed by calculating the Medication Possession Ratio, and satisfaction and fulfilment of expectations using the EXPRESAR group questionnaire.
ResultsIn 346 patients, those who were satisfied/very satisfied with efficacy and tolerability were ≥80% and 64.4%, with no differences between weekly, biweekly or monthly administration regimens. Regarding the fulfilment of expectations, 59.9% considered the effect of the treatment greater than expected and 52.6% reported lower/much lower than expected discomfort; the latter percentage was higher in patients with monthly administration (P = .049). The percentages for nonadherence were 15.6% (discomfort greater than expected), 18.5% (expected discomfort) and 11.1% (lower than expected or no discomfort) (P = .189).
ConclusionsSatisfaction and fulfilment of expectations were high. Fulfilment of expectations of tolerability was better with monthly administration, which could contribute to better adherence.
En el estudio ARCO, la adherencia al biológico subcutáneo en pacientes con artritis reumatoide fue mejor con administración mensual. Evaluamos si satisfacción y cumplimiento de expectativas pueden relacionarse con la adherencia al tratamiento.
Pacientes y métodosLa adherencia se evaluó calculando el Índice de Posesión de Medicación, y satisfacción y cumplimiento de expectativas mediante el cuestionario EXPRESAR.
ResultadosEn 346 pacientes, los porcentajes satisfechos/muy satisfechos con eficacia y tolerabilidad fueron ≥80% y 64.4%, sin diferencias entre pautas semanal, cada dos semanas o mensual. Sobre el cumplimiento de expectativas, el 59.9% consideró el efecto del tratamiento mayor al esperado, y el 52.6% las molestias bastante/mucho menores a las esperadas, porcentaje mayor en pacientes con administración mensual p = 0,049. Los porcentajes de no-adherencia fueron 15.6% (molestias mayores a las esperadas), 18.5% (molestias esperadas) y 11.1% menores/sin molestias p = 0,189.
ConclusionesLa satisfacción y el cumplimiento de expectativas fueron altos. El cumplimiento de expectativas de tolerabilidad fue mejor con administración mensual, lo que podría contribuir a una mejor adherencia.
Rheumatoid arthritis (RA) requires chronic treatment, and adherence to this is important to gain the best therapeutic results.1 Nevertheless, adherence to anti-rheumatic medication is sub-optimum,2 and the lack of adherence has been associated with the type of medication, the administration regime, the psychological characteristics of patients or beliefs respecting the medication.3,4
One aspect that has been studied less is the relationship between adherence and the expectations and degree of satisfaction of patients regarding treatment. In the study “Adherence of Rheumatoid arthritis patients to subCutaneous and Oral drugs” (ARCO), of 364 patients with RA, we described how, during the first 14 months of treatment, 14.3% of these patients did not adhere to the prescribed subcutaneous (SC) biological drug,5 and that the percentage of non-adherent patients was lower in those for whom administration was monthly (6.4%) than it was for patients treated every fortnight (14.4%) or weekly (17.4%). This work has two objectives: 1) to describe the degree of satisfaction and fulfilment of expectations of the patients with their treatment, and 2) to evaluate whether there are differences between administration regimes that may contribute to a higher level of adherence, apart from the regime itself.
MethodsARCO was a multicentre retrospective study in patients ≥18 years old with RA who had started taking a SC biological therapy 12–18 months before the study. Adherence to the SC biological drug (the primary objective) was evaluated by calculating the Medication Possession Ratio [MPR], considering a MPR ≤ 80% as non-adherence. The information about the design and main findings has already been published.5 The patients also filled out a questionnaire developed by the EXPRESAR6 group on their satisfaction and expectations. This evaluated their degree of satisfaction with the efficacy and tolerability of the treatment on a Likert scale (from “very dissatisfied” to “very satisfied”), and fulfilment of their expectations was assessed from “much better” to “much worse than expected”.
In this work we describe the degree to which patients were satisfied with the treatment and how far it fulfilled their expectations. We study whether there are any differences between administration regimes for the SC biological drug, as well as if the percentage of non-adherence differs according to the degree of satisfaction and the fulfilment of expectations. The patients were classified according to the administration regime of the SC biological drug they were taking, as weekly, fortnightly or monthly. Regarding their level of satisfaction they were divided into “satisfied/very satisfied” or “neutral/dissatisfied”, and respecting the fulfilment of their expectations, they were divided into “much/quite a lot better than expected”, and “more or less as expected” or “quite a lot/much less than expected”. The descriptive data are shown with core tendency measurements, scatter and percentages. The differences between percentages were analysed using the chi-squared test or Fischer’s exact test.
ResultsFrom May 2014 to September 2015 364 patients were recruited (54.9 years old [SD: 12.5]; 77.5% were women, with median duration of RA of 7.8 years [interquartile range: 3.4–15.7]). The interval between administrations of the SC biological drug was weekly (n = 161, 44.2%), fortnightly (n = 140; 38.5%) or monthly (n = 63; 17.3%). As well as the SC biological drug, 77.2% were also being treated with oral anti-rheumatic drugs and 31.6% were taking non-biological SC anti-rheumatic drugs. The 346 patients who filled out the EXPRESAR group questionnaire had similar characteristics.
A very high percentage of the patients were satisfied/very satisfied with the efficacy of the treatment (close to or higher than 80% in all aspects except for foot pain [Table 1]). 64.4% considered themselves to be satisfied/very satisfied with tolerability (side effects or discomfort). The percentages of patients who were satisfied/very satisfied with efficacy (question 1) were very similar in the patients with weekly, fortnightly and monthly administration, while the percentages who were satisfied/very satisfied with the side effects or discomfort were 63.9%, 61.2% and 72.9%, respectively (P = .176). The percentage of patients who did not adhere to their SC biological drug regime amounted to 14.7% of those who were satisfied with efficacy and 8.3% of those who were neutral/dissatisfied (P = .339). The percentages of non-adherence were very similar respecting side effects (13.1% vs. 15.4%, respectively; P = .504).
Percentages of patients who answered that they were satisfied or very satisfied in reply to the EXPRESAR group questionnaire.
| No. (%) satisfied or very satisfied | |
|---|---|
| 1. Do you feel satisfied with its efficacy? | 301 (86.2) |
| 2. Do you feel satisfied with how it is controlling symptoms? | 301 (86.2) |
| 3. Do you feel satisfied with the treatment regarding pain in your joints in general? | 291 (83.1) |
| 4. Do you feel satisfied with the treatment regarding pain in your hands? | 274 (78.8) |
| 5. Do you feel satisfied with the treatment regarding pain in your feet? | 239 (68.9) |
| 6. Do you feel satisfied with the treatment regarding the reduction of joint inflammation? | 297 (85.1) |
| 7. Do you feel satisfied with the side effects or discomfort (such as dizziness or stomach pain)? | 222 (64.4) |
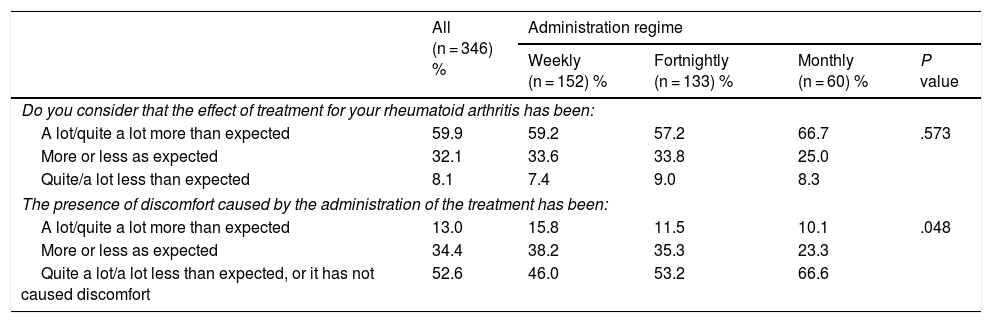
Table 2 shows the degree to which expectations were fulfilled. The majority stated that the effect had been greater than expected, without differences here between different administration regimes. The percentages of non-adherence were 15.5% among those who considered the effect to be greater than expected, 12.6% among those considered it to be similar and 10.7% of those who described it as less than expected (P = .667).
Fulfillment of expectations in terms of efficacy and tolerability according to the administration regime of the SC biological drug.
| All (n = 346) % | Administration regime | ||||
|---|---|---|---|---|---|
| Weekly (n = 152) % | Fortnightly (n = 133) % | Monthly (n = 60) % | P value | ||
| Do you consider that the effect of treatment for your rheumatoid arthritis has been: | |||||
| A lot/quite a lot more than expected | 59.9 | 59.2 | 57.2 | 66.7 | .573 |
| More or less as expected | 32.1 | 33.6 | 33.8 | 25.0 | |
| Quite/a lot less than expected | 8.1 | 7.4 | 9.0 | 8.3 | |
| The presence of discomfort caused by the administration of the treatment has been: | |||||
| A lot/quite a lot more than expected | 13.0 | 15.8 | 11.5 | 10.1 | .048 |
| More or less as expected | 34.4 | 38.2 | 35.3 | 23.3 | |
| Quite a lot/a lot less than expected, or it has not caused discomfort | 52.6 | 46.0 | 53.2 | 66.6 | |
SC: subcutaneous.
Half of the patients considered that the discomfort caused by administration were quite or much less than expected or were absent, and this percentage was higher in the patients with monthly administration (P = .049, Table 2). The percentages who said that it caused no discomfort were 17.8% (weekly administration), 29.3% (fortnightly) and 35.0% (monthly, P = .013). The percentages of non-adherence were 15.6% in those with more discomfort than expected, 18.5% in those with an expected level of discomfort and 11.1% in those with less discomfort than expected or without discomfort (P = .189).
DiscussionIn the ARCO study we reported a higher rate of adherence to the SC biological drug with monthly administration.5 In the current study we examine possible differences in greater depth from the viewpoint of patient experience which can be associated with adherence, apart from the administration regime itself. Although no significant differences were found in adherence according to degree of satisfaction or the fulfilment of expectations, and with the limitation of sample size, the information supplied by the patients contains relevant data.
There was a very high level of satisfaction with the therapeutic effect, without differences between administration regimes. This finding fits with the high level of efficacy of the biological drugs, which is similar for all of them,7 and it also agrees with the findings of a recent survey in our field of 592 patients, who expressed a level of satisfaction with the efficacy of the SC biological drug of ≈80%.8 Although there was less satisfaction with the side effects, it still attained a high level (64%), with slight differences that were not significant between administration regimes. This aspect is relevant as these patients generally require chronic treatment.
The expectations patients feel when they start a treatment and whether they are fulfilled are also relevant.9 In our study the percentage who stated that they felt less discomfort than expected or no discomfort at all was highest in the patients with a monthly dose requiring fewer injections. The study cannot determine whether this finding is due to the lower number of injections in itself or to better tolerability of the drug. The percentage of non-adherence stood at 11.1% in the patients with less discomfort than expected or no discomfort. This figure is slightly lower than the 18.5% and 15.6% of non-adherence in patients with “expected or more than expected discomfort”. The doctor–patient relationship was also said to be an important factor in improving adherence: expertise regarding the disease and feeling expectations with the patients about the effect and tolerability of the treatment may lead to benefits in terms of better adhesion and clinical results.10
Our study has several limitations. The sample size (n = 346) lacks the power necessary to find significant differences between adherence percentages, although it does suggest that patients whose expectations of tolerability were fulfilled displayed better adherence. The majority of the patients said they were satisfied or very satisfied with the efficacy of the treatment, hindering evaluation of the percentages of adherence according to this variable. At the time the study was undertaken, the only SC biological drugs were anti-TNF alpha; there are now SC biological drugs with other mechanisms of action. Given all of these considerations, exploring patient-related aspects (belief in the medication, expectations and experience) is of interest in taking steps to improve adherence and clinical results,11 and this work offers interesting information in these fields.
To conclude, the patients with RA who were treated with SC biological drugs expressed a high level of satisfaction and fulfilment of their expectations respecting the efficacy of the treatment. Although the percentage of patients who were satisfied with its tolerability was also high, it was lower than it was for efficacy. The fulfilment of expectations seems to be higher with a monthly injection regime, so that this, together with the fact that fewer SC injections are needed, may contribute to adherence to treatment with the drug.
FinancingThe ARCO study was financed by Merck Sharp & Dohme, Spain.
Conflict of interestsLuis Cea-Calvo is a full-time employee of Merck Sharp & Dohme in Spain. The other authors have no conflict of interests to declare.
We would like to thank all of the researchers and patients who took part in filling out the questionnaires that were necessary to obtain this information.
Please cite this article as: Calvo-Alén J, Vela P, Bustabad S, Maceiras F, Carmona L, Cea-Calvo L. Satisfacción, cumplimento de expectativas y adherencia al fármaco biológico subcutáneo en pacientes con artritis reumatoide. Estudio ARCO. Reumatol Clin. 2020;16:116–119.