To develop a joint proposal for screening criteria of interstitial lung disease (ILD) in patients with rheumatoid arthritis (RA) and vice versa, which serves as a guidelines in patient referral between the Rheumatology and Pneumology departments to early detection of these patients.
MethodsA systematic literature review was carried out on the risk factors for the development of ILD in RA patients, and for the referral criteria to Rheumatology for suspected early RA. Based on the available evidence, screening criteria were agreed using the Delphi method by a panel of pneumologists and rheumatologists with expertise in these pathologies.
ResultsScreening criteria for ILD in patients with RA and for the early detection of RA in cases with ILD of unknown etiology have been developed. In both cases, a detection strategy was based on clinical risk factors. Recommendations also included the complementary tests to be carried out in the different clinical scenarios and on the periodicity that screening should be repeated.
ConclusionA selective screening strategy is recommended for the first time in the early diagnosis of patients with ILD-RA. This multidisciplinary proposal aims to solve some common clinical questions and help decision-making, although its usefulness to identify these patients with good sensitivity must be confirmed in a validation study.
Elaborar una propuesta multidisciplinar de criterios de cribado de enfermedad pulmonar intersticial difusa (EPID) en pacientes con artritis reumatoide (AR) y a la inversa, que sirvan de referencia en la derivación entre los Servicios de Reumatología y Neumología para la detección precoz de estos pacientes.
MétodosSe revisó de forma sistemática la literatura sobre factores de riesgo para el desarrollo de EPID en la AR, la utilidad de los distintos métodos diagnósticos para su identificación en pacientes con AR y las diferentes propuestas de criterios de derivación a Reumatología por sospecha de AR precoz. Basándose en la evidencia disponible y en su experiencia clínica, un comité científico formado por 2 reumatólogos y 2 neumólogos propuso unos criterios de cribado que fueron evaluados mediante el método Delphi por un panel de 7 neumólogos y 7 reumatólogos. Todos los participantes eran expertos en esta patología.
ResultadosSe han elaborado unos criterios para el despistaje de EPID en pacientes diagnosticados de AR, y unos criterios para la detección precoz de AR en casos de EPID de causa no filiada. Se incluyen también propuestas sobre las pruebas complementarias a realizar en los diferentes escenarios clínicos considerados y sobre la periodicidad con la que debe repetirse el cribado.
ConclusionesSe propone por primera vez una estrategia de cribado selectivo para el diagnóstico precoz de los pacientes con EPID-AR. Esta propuesta pretende resolver algunos interrogantes clínicos habituales y facilitar la toma de decisiones. Los criterios propuestos deben ser evaluados en futuros estudios de validación.
Interstitial lung disease(ILD) is one of the most common and severe extra-articular manifestations of rheumatoid arthritis (RA)1–4. In more than three-quarters of patients, ILD occurs after a diagnosis of RA, usually within the first 5–10 years of disease progression1–7. In these cases, it is not uncommon for it to be diagnosed late, as it is often asymptomatic or paucisymptomatic in its early stages1–7. In fact, when the entire RA population was screened for ILD by chest CT scan, a not insignificant percentage of subclinical disease was detected (between 11.9% and 55.7%), which confirms that this complication is underdiagnosed8,9. Less frequently, ILD occurs at onset of RA or precedes joint manifestations by months or years. In the latter scenario, ILD-RA is often confused with idiopathic forms, despite their different prognosis and treatment1–3,10,11.
Although a significant improvement in its prognosis has been observed12, ILD is still the second leading cause of premature death in RA, after cardiovascular complications1–4,10. Despite its frequency and severity, there are currently no recommendations on screening for this complication, in either the initial assessment or in the follow-up of RA patients.
Based on the above, and to respond to this clinical need, the objective of the RA-EPIDSER project was to draft a multidisciplinary proposal for screening criteria that would enable the early identification of patients with ILD-RA.
Material and methodsA systematic review of the literature and consensus techniques (Delphi method with a minimum of two rounds) were used to determine the degree of consensus of experts based on their clinical experience and scientific evidence.
Project stagesSelection of expertsFirst, the AR-EPIDSER project working group was formed, which comprised a scientific committee (SC) formed by two rheumatologists (JN and SC) and two pulmonologists (MA and CV), and a panel of experts formed by seven rheumatologists (GB, GC, IC, HC, MML, NMV, and AO), and seven pulmonologists (OA, ECJ, JMGR, BLM, ARP, JARP, and CSA). All were selected based on their experience in the area of interest and their scientific merits, and for the greatest geographical representativeness: the rheumatologists obtained the highest score in a public call for candidates among the members of the Spanish Society of Rheumatology (SER). The pulmonologists were selected by the ILD area of the Spanish Society of Pneumology and Thoracic Surgery (SEPAR), according to the following criteria: belonging to a SEPAR-accredited ILD unit and representing the different levels of accreditation.
Systematic literature reviewThree systematic literature reviews were conducted: of the risk factors for developing ILD in RA, of the usefulness of the different diagnostic methods for screening for ILD in RA, and of the different proposals for criteria for referral to rheumatology for suspected early RA with sensitive literature search strategies (using free terms and Mesh terms) in Medline, Embase, and Cochrane Library.
Evidence synthesis was peer reviewed. The reviewers (IC, RDS, SGC, JM, and LSF) were selected based on their training in evidence-based medicine and their previous experience in writing systematic reviews; they were also supported by the SER’s Research Unit (RU).
The methodology used is shown in Appendix B Annex A of the supplementary material. The research questions in PICO format and the search strategies were defined by the SC, members of the SER’s RU, and the reviewers overseeing the evidence synthesis.
Proposed screening criteriaBased on the evidence found and their clinical experience, the SC members agreed, with the support of a methodologist (DS) from the RU, on two proposals for selective screening: criteria for ILD screening in patients diagnosed with RA, and criteria for the early detection of RA in patients with ILD of undetermined or unidentified cause. Proposals were also included for the complementary tests to be performed in the different clinical scenarios considered and the frequency of repeat screening.
These proposals were assessed by the members of the expert panel by means of a two-round Delphi in electronic format in which they scored their degree of agreement with each of the items according to the following scale: 1 = strongly disagree; 2 = somewhat disagree; 3 = neither agree nor disagree; 4 = somewhat agree; and 5 = strongly agree. In addition to rating the proposals, the panellists could also make suggestions for modifications or propose additional criteria in a free text field.
The degree of agreement for each item was defined as the percentage of panellists who awarded a score ≥4, and the degree of disagreement as the percentage of scores ≤2. Consensus was established in cases with scores ≥4 from at least 75% of the panellists.
The SC assessed the criteria on which there was no consensus in the first Delphi round, as well as proposals for additional criteria. This resulted in a second version of the document that incorporated, for each item to be assessed, summary parameters of the group's response and the comments made in the first round. This new version was sent to the panellists for them to compare their initial assessment with that of the group and score the items again, using the same scale as in the first round.
Throughout the process, the anonymity of the expert panel members was ensured. They all completed the successive rounds of the Delphi method, conducted between July and October 2020.
ResultsProposed screening criteria for ILD in patients diagnosed with RAThe main risk factors for developing ILD in RA identified in the systematic literature review are shown in Table 1. Based on the evidence found and on their clinical experience, the SC members decided to propose screening for ILD in three different scenarios:
- 1
In patients with respiratory symptoms (cough and/or dyspnoea) of more than 3 months’ duration.
- 2
In patients in whom “Velcro-like” crackles are detected on respiratory auscultation, even if asymptomatic.
- 3
In patients without respiratory symptoms and with normal respiratory auscultation, screening will be based on the score obtained according to their number of risk factors for developing this complication.
Main risk factors for the development of interstitial lung disease in rheumatoid arthritis.
| Risk factors | Level of evidence |
|---|---|
| Male sex15,16 | 2+ |
| Advanced age and late onset of disease15,17–20 | 2++ |
| Duration of rheumatoid arthritisa,15,18,21 | 2+ |
| Smoking17,18,20,22 | 2++ |
| Moderate or high sustained RA activity according to DAS28 scores23 | 2++ |
| Rheumatoid factor positive15,18,24 | 2++ |
| Anti-cyclic citrullinated peptide antibody (ACPA) positive, especially at high titres15,18,24–26 | 1+ |
| Biomarkers under evaluation | |
| Anti-carbamylated protein (anti-CarP) antibodies27 | 3 |
| Extracellular matrix metalloproteinase 7 (MMP-7)28–30, interferon gamma-inducible protein-10 [IP-10] or CXCL1028, interleukin 1829, 90 and 70-kDa heat shock protein (HSP90/70)31 | 2– |
| Genetic biomarkers | |
| MUC5B gene mutations32 | 2+ |
| Mutations in telomerase genes that cause accelerated telomere shortening33,34 | 2+ |
DAS28: Disease activity score-28; MUC5B: mucin 5B; RA: rheumatoid arthritis.
In the latter scenario, the following risk factors were included in the SC’s initial proposal (see Appendix B Annex B of the supplementary material): advanced age (with 2 different cut-off points: ≥55 years or ≥60 years), male sex, smoking history, disease duration >5 years, moderate or high maintained RA disease activity, and rheumatoid factor (RF), and/or anti-cyclic citrullinated peptide antibody (ACPA) positivity and titre. Each of these factors was given a score, patients with a total score ≥4 points were considered eligible for screening. The relative contribution or weight of each of the variables included and the optimal cut-off point that, when applied to these patients, would identify those with a high likelihood of developing ILD, was established based on the available evidence and the clinical judgement of the SC members.
Appendix B Annex 2 also show the results of the assessment of the Delphi rounds. In the first round, consensus was reached for screening in all three proposed scenarios.
However, in the proposal for screening in asymptomatic patients with normal auscultation, there were disagreements on the cut-off point and score given to age, on how to quantify cumulative smoking, on whether or not to keep disease duration at a cut-off point >5 years and on setting the threshold for the total score at 4 points. Although there was agreement on the score assigned to male sex, following the SC's assessment of the comments made by some of the panellists, it was decided to include it in the second round as well.
In addition to proposing alternatives to reach consensus on these 4 items in the second Delphi round, the SC also included a family history of ILD in the risk factors to be considered, at the suggestion of the panellists.
No consensus was reached after the second round on these items either, except to accept disease duration at >5 years (see Appendix B Annex B) and to set the threshold for the total score at 5 points. Although not initially planned, it was decided to re-evaluate the remaining factors in a third Delphi round, but consensus could not be reached, except for on the inclusion of a family history of ILD. Finally, the SC chose to include the option with the highest degree of agreement in the final version of the proposal. This final version is shown in Table 2.
Proposed screening criteria for interstitial lung disease screening in patients diagnosed with rheumatoid arthritis.
| ILD screening will be performed in these three clinical situations | Degree of agreementa |
|---|---|
| 1. Patients with respiratory symptoms (cough and/or dyspnoea) of more than 3 months’ duration | 100% (1st Delphi round) |
| 2. Patients in whom “Velcro-like” crackles are detected on respiratory auscultation, even if asymptomatic | 100% (1st Delphi round) |
| 3. In patients without respiratory symptoms and with normal respiratory auscultation, screening will be done according to the score obtained for the number of risk factors present for developing this complication | 78.6% (2nd Delphi round) |
| Any patient scoring ≥5 points will be considered eligible for screening |
| Set of variables and proposed score for each of the variables for overall calculation | Score |
|---|---|
| Age ≥ 60 years | 2b |
| Male sex | 1b |
| History of smoking (active or ex-smoker) | |
| ≤ 20 packs/year: 2 points | 2b |
| >20 packs/year: 3 points | 3b |
| Disease duration > 5 years | 1c |
| Persistent moderate-high disease activity: DAS28-VSG average > 3.2 from diagnosis of the disease in RA onset (time from diagnosis ≤ 12 months) or DAS28-VSG > 3.2 for a minimum of 6 months in established RA | 1c |
| Serology (only the criterion with the highest weighting is counted towards the total score) | |
| RF positive > 3 times above the ULN | 1c |
| ACPA positive ≤ 3 times above the ULN | 2c |
| ACPA positive > 3 times the ULN | 3c |
| Family history of ILD | 1c |
| How to screen | |
|---|---|
| In patients with cough and/or dyspnoea of more than 3 months’ duration, chest X-ray and RFT including spirometry and DLCO. Depending on the result, HRCT should be considered* | 71.4% (3rd Delphi round; no consensus) |
| In patients with “Velcro-like” crackles on respiratory auscultation, an HRCT scan should be performed directly | 100% (1st Delphi round) |
| In patients with no respiratory symptoms and normal respiratory auscultation, with risk factor score of 5−6, chest X-ray, and RFT including spirometry and DLCO should be performed initially. Depending on the result, HRCT should be considered* | 78.6% (2nd Delphi round) |
| If the score is ≥ 7, perform HRCT directly | 85.7% (2nd Delphi round) |
| * If RFT is unavailable or there is an excessive waiting list, consider performing HRCT directly to speed up diagnosis. Performing HRCT directly does not exclude also performing a RFT to assess the severity of the ILD | 71.4% (3rd Delphi round) |
A third round was required for the cut-off point and score for age, the score assigned to male sex and the way of assessing smoking history, no consensus was reached: degree of agreement of 71.4% for sex and smoking. For age it was 85.7% for a cut-off point of 60 years, and 57.1% for the assigned score.
The SC made specific proposals for each of the three proposed scenarios, which are detailed in Table 2.
In scenario one (patients with cough and/or dyspnoea of more than 3 months’ duration), consensus was not reached; the degree of agreement after three Delphi rounds was 71.4%. The main reason given by the panellists who disagreed with the SC proposal was that the screening test should directly be a high-resolution chest CT (HRCT).
If unavailable or there is an excessive waiting list for respiratory function tests (RFT), the possibility of direct HRCT is left open to speed up diagnosis. Direct HRCT does not exclude the possibility of also performing RFTs to assess the severity of the ILD.
Proposed criterion for RA screening in patients diagnosed with ILD of undetermined causeBased on the evidence and their clinical experience, the SC members decided in their initial version to propose referral to rheumatology to investigate the presence of RA in patients with ILD of undetermined cause presenting with one or more of the following criteria:
- 1
ACPA positivity.
- 2
RF positivity:
- -
At any titre in those under 65 years of age.
- -
In those older than 65 years, 2 cut-off points were proposed for assessment by the panellists: titres ≥2 or ≥3 times above the upper limit of normal (ULN).
- -
- 3
The presence for at least 4 weeks of one or more of the following joint manifestations:
- -
Pain in metacarpophalangeal or metatarsophalangeal joints, measured by lateral joint compression test or squeeze test.
- -
Morning joint stiffness lasting more than 30 min.
- -
Joint swelling in ≥2 or ≥3 joints (these two options were proposed for assessment by the panellists).
- -
After the first Delphi round, consensus was reached on all criteria except RF (see Appendix B Annex B). Regarding joint swelling, of the two options assessed, only swelling in two or more joints achieved consensus.
Of the different alternatives that were proposed for the RF criterion, the only one that reached consensus after the second Delphi round was RF positivity at titres ≥2 at ULN, irrespective of patient age (Appendix B Annex B). The final version of the criteria is shown in Table 3.
Proposed criteria for screening for rheumatoid arthritis in patients diagnosed with interstitial lung disease of undetermined cause.
| [0,1–2]Patients with ILD of unidentified aetiology should be referred to rheumatology when presenting with one or more of the following criteria | |
|---|---|
| Degree of agreementa | |
| Anti-cyclic citrullinated peptide antibody (ACPA) | 100% (1st Delphi round) |
| Rheumatoid factor positive at titres ≥ 2 of ULN | 92.9% (2nd Delphi round) |
| The presence for at least 4 weeks of one or more of the following joint manifestations: | 92.9% (1st Delphi round) |
| Pain in the metacarpophalangeal or metatarsophalangeal joints, measured by side-lying joint compression or squeeze test | 85.7% (1st Delphi round) |
| Morning joint stiffness of more than 30 min’ duration | 92.9% (1st Delphi round) |
| Joint swelling in ≥ 2 joints | 78.6% (1st Delphi round) |
ILD: diffuse interstitial lung disease; ULN: upper limit of normal.
Consensus was reached in cases with scores ≥ 4 awarded by at least 75% of the panellists.
Proposals for the frequency of ILD screening in patients with RA are shown in Table 4, while those for the frequency of RA screening in patients diagnosed with ILD of undetermined cause are shown in Table 5.
Frequency of screening for interstitial lung disease in patients with rheumatoid arthritis.
| [0,1–2]Frequency of screening | |
|---|---|
| Degree of agreementa | |
| In the follow-up of patients with RA, auscultation should be performed at least once a year, in addition to specific questioning about the presence of respiratory symptoms and assessment of risk factors for the development of ILD according to the scoring system shown above | 92.9% (1st Delphi round) |
| If “Velcro-like” crackles or respiratory symptoms (cough and/or dyspnoea of more than 3 months’ duration) are detected during follow-up, screening tests should be repeated according to the above recommendations, regardless of whether they were negative in the past | 100% (1st Delphi round) |
| In asymptomatic patients with normal respiratory auscultation with a total score ≥ 5 and negative screening test result, screening should be repeated once a year with spirometry and DLCO | 85.7% (1st Delphi round) |
DLCO: Diffusing capacity of the lungs for carbon monoxide; ILD: diffuse interstitial lung disease; RA: rheumatoid arthritis.
Frequency of screening for rheumatoid arthritis in patients diagnosed with interstitial lung disease of undetermined cause.
| [0,1–2]Frequency of screening | |
|---|---|
| Degree of agreementa | |
| In the follow-up of these patients, they will be specifically asked about the presence of joint symptoms and a musculoskeletal examination will be performed every 6 months for the first two years following diagnosis of ILD, and annually thereafter | 92.9% (1st Delphi round) |
| RF and ACPA tests will be repeated annually for the first 5 years after the diagnosis of ILD. Thereafter, they will be repeated at medical discretion according to the patient’s progress | 78.6% (1st Delphi round) |
ACPA: Anti-cyclic citrullinated peptide antibody; ILD: Diffuse interstitial lung disease; RF: Rheumatoid Factor.
ILD is one of the most frequent and severe extra-articular manifestations of RA and is currently the second leading cause of premature death from RA. Median survival from diagnosis of ILD varies between 2.6 and 8.1 years13.
However, unlike in systemic sclerosis, in which ILD is also the first or second cause of death14, in RA there are no recommendations for screening for this complication, despite the fact that it is underdiagnosed8,9 and that it often goes unnoticed until advanced stages, when the extent of fibrotic changes limits therapeutic possibilities and survival (it should be noted that interstitial pneumonia is the most frequent histopathological pattern in RA).
To correct this situation and improve the prognosis and quality of life of patients with ILD-RA, the SER promoted the AR-EPIDSER study, in which a group of rheumatology and pulmonology experts was formed to jointly develop criteria for the early identification of patients with ILD-RA to serve as a benchmark for referral of these patients to rheumatology and pulmonology services. This multidisciplinary proposal aims to resolve some common clinical questions and facilitate decision-making in different clinical scenarios. In the absence of evidence to support the cost-effectiveness of universal screening, selective screening proposals were chosen based on the patients’ clinical risk.
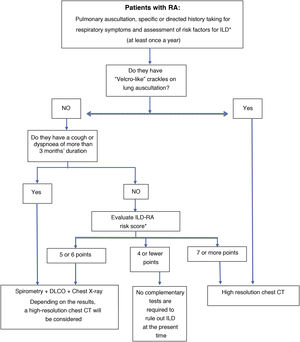
Regarding the criteria for when to investigate for ILD in patients diagnosed with RA, the desired consensus was reached in most of the situations considered, and a screening algorithm useful in clinical practice was proposed (Fig. 1). As a limitation, in the situations referring to screening in asymptomatic patients with normal auscultation, consensus was not reached on some of the items included in the scoring system, specifically on the cut-off point and score given to age, on the score assigned to male sex, and on how cumulative smoking should be quantified. Consensus was not reached on how to screen patients with respiratory symptoms of more than 3 months' duration either. In these cases, the SC opted to include in the final version of the proposal the option with the highest degree of agreement, which for most of these criteria was 71.4%, i.e., very close to consensus. Sixty was finally selected as the cut-off point for age, which, in addition to being the most voted for, is also a prognostic factor associated with higher mortality13–35.
Proposed screening algorithm for interstitial lung disease (ILD) screening in patients diagnosed with rheumatoid arthritis (RA).
* See risk factors and their score in Table 2.
? If screening tests are negative, screening will be repeated once a year by spirometry + DLCO.
To prevent this proposal from rapidly becoming obsolete, the SC considered the option of incorporating the new biomarkers under investigation into the scoring system27–31, fundamentally anti-carbamylated protein (anti-CarP) antibodies. A correlation has been shown to exist between levels of all anti-CarP specificities (foetal bovine serum, fibrinogen, and chimeric fibrin/filaggrin homocitrullinated peptide) and the presence of ILD, after adjustment for other known risk factors27. Promising preliminary results have also been obtained with serum levels of extracellular matrix metalloproteinase 7 (MMP-7), interferon gamma-inducible protein-10 (IP-10) or CXCL10, interleukin 18, and 90 and 70-kDa heat shock protein (HSP90/70)27–31. Finally, it was decided not to include them as they are not yet available in clinical practice and have not yet been shown to have a higher predictive value for the development of this complication than ACPA. Genetic biomarkers, including mutations in the MUC5B gene32 and mutations in telomerase genes that cause accelerated telomere shortening33,34, are not yet available in clinical practice. Nevertheless, and as an indirect way of recognising its future usefulness and the importance of genetic predisposition in the development of ILD in a subgroup of patients, it was agreed to include a family history of ILD as a risk factor to consider in the algorithm.
An important message that the authors of both specialties want to highlight in this proposal is the usefulness and importance of respiratory auscultation in the rheumatology consultation for ILD screening. The presence of “Velcro-like” crackles correlates well with the presence of pulmonary fibrosis36–38. In addition, two studies have shown that conventional respiratory auscultation has acceptable diagnostic accuracy for the detection of ILD when compared with that obtained with a digital electronic stethoscope36,37.
Of the tests to screen for ILD, the scientific committee decided not to include lung ultrasound for the time being, considering that it is a technique that has not yet been sufficiently validated to diagnose this complication. A recent systematic review by the Outcome Measures in Rheumatoid Arthritis Clinical Trials (OMERACT) group confirms its good face and content validity, and its feasibility, but it does not yet have criterion validity, reliability or, above all, sensitivity to change39. Limitations still to be resolved include the lack of a clear definition of normality, the absence of agreed definitions of the lesions assessed, and the lack of standardisation of the technique, image acquisition, and the number of areas to explore (which in the studies varies from 10 to 72). There was also no consensus on the quantification of pneumopathy, on the scoring system, or on the optimal probe to use (3–3.5 to 5, or 7.5 MHz). Likewise, there is a lack of longitudinal follow-up studies and studies assessing their ability to diagnose ILD in early stages (subclinical ILD)39.
Another important clinical scenario that considers these recommendations and of which pulmonologists should be made aware is the pulmonary debut of RA. As already mentioned, in 10%–14% of cases of ILD-RA, pulmonary involvement precedes joint manifestations by months or years1–3,10,11. In these cases, early referral to the rheumatologist by the pulmonologist will make it possible not to delay diagnosis and to initiate remission-inducing treatment as soon as the clinical diagnosis of the disease is established, thus taking advantage of the so-called “window of therapeutic opportunity”, understood as a period of time very early in the onset of the disease in which, with appropriate intervention, we can modify the course of the disease and avoid joint damage and associated complications.
Therefore, the initial assessment of all patients with ILD should include a detailed clinical history and a thorough physical examination to investigate the presence of extrapulmonary manifestations, together with an analytical study that includes at least the erythrocyte sedimentation rate (ESR), C-reactive protein and testing for RF, ACPA, antinuclear antibodies, and myositis-specific autoantibodies, as recommended by the European and American societies of Pneumology40. We also include in our proposal the screening for RA in the follow-up of patients with ILD of undetermined aetiology, with the frequency described in the present study.
In this clinical scenario, the predictive value of ACPA appears to be superior to that of RF41–44. Positive RF is not pathognomonic of RA, having been described in 5%–10% of the healthy population, especially in elderly patients, although generally at low titres. Hence, in the proposal, it was decided to consider only RF titres ≥2 at ULN. The joint manifestations included, based on which to suspect early arthritis, are easily identifiable by a non-rheumatologist. Their validity has been contrasted in population-based studies, including one conducted in Spain45,46.
The number of experts participating in the Delphi is within the range accepted as valid and all of them completed their participation during the successive rounds. The threshold set at 75% to define consensus and the number of Delphi rounds are also within the parameters considered adequate47,48. Despite following a rigorous methodology, this proposal has several limitations. The main limitation, in addition to the aforementioned lack of consensus for some of the criteria, is that this proposal is only an expert consensus, the usefulness of which has not yet been tested in validation studies. Moreover, given that the panel was only a national one, it may not be possible to extrapolate these criteria to other health systems.
ConclusionsCurrent evidence flags up the need for a common strategy between rheumatologists and pulmonologists to improve the early diagnosis of patients with ILD-RA, given its prognostic and therapeutic implications. To respond to this need, for the first time a multidisciplinary group of rheumatologists and pulmonologists have developed a consensual proposal for selective screening criteria as an aid to clinical decision-making. Its usefulness in identifying these patients in clinical practice will need to be confirmed in future validation studies.
Ethical responsibilitiesProtection of humans and animalsThe authors declare that no experiments on humans or animals have been conducted for this research.
Data confidentialityThe authors declare that they have followed their centre's protocols on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
AuthorshipAll authors have contributed substantially to: a) the conception and design of the study and analysis of the data; b) drafting the article or critical revision of the intellectual content; and c) the final approval of the submitted version.
FundingThis project was funded by Bristol Myers Squibb, who were not involved in the design of the study, the conduct of the study, or drafting of this article.
Conflict of interestsThe authors have no conflict of interests to declare.
Co-principal authors: Javier Narváez and Myriam Aburto.
Please cite this article as: Narváez J, Aburto M, Seoane-Mato D, Bonilla G, Acosta O, Candelas G, et al. Criterios de cribado de la enfermedad pulmonar intersticial difusa asociada a la artritis reumatoide: propuesta de expertos basada en metodología Delphi. Reumatol Clin. 2023;19:74–81.