Sjögren's Syndrome (SS) is an autoimmune disease with a wide spectrum of clinical manifestations that can have an important impact on the patient's quality of life. To make an objective evaluation of the components of the disease, clinimetric tools such as the ESSPRI have been designed. The objective of this study is to adapt this scale to the Spanish language.
Materials and methodsThis is a cross-sectional study to validate clinimetric scales, carried out in Cali, Colombia. A translation of the original English version of ESSPRI into Spanish was made and applied to patients with SS, as well as PROFAD and ESSDAI, as an activity marker. The reliability index of the questionnaire in Spanish with Cronbach's alpha coefficient and Spearman's correlation coefficient were calculated to compare the scales. Demographic, clinical and laboratory characteristics were also evaluated.
ResultsESSPRI, PROFAD and ESSDAI were applied to 42 patients with SS, 97.62% were women. The average result of the ESSPRI was 5.8 (± 4.6), with a reliability coefficient of .8034 and a correlation with PROFAD of .5800 (p=.0001), and of -.0848 (p=.593) with ESSDAI.
Discussion and conclusionsReliability with the applied version of ESSPRI in Spanish was adequate. A discrepancy was found between this scale and ESSDAI, which highlights the importance of applying both tools to ensure objective monitoring of disease control and its impact on the quality of life of patients with SS.
El Síndrome de Sjögren (SS) es una enfermedad autoinmune con un amplio espectro de manifestaciones clínicas que puede generar un importante impacto en la calidad de vida del paciente. Para hacer una evaluación objetiva de los componentes de la enfermedad, se han diseñado herramientas clinimétricas como el ESSPRI. El objetivo de este estudio es hacer una adaptación de esta escala al idioma castellano.
Materiales y métodosEs un estudio transversal de validación de escalas clinimétricas, llevado a cabo en Cali, Colombia. Se realizó una traducción de la versión original en inglés del ESSPRI al castellano y se aplicó a pacientes con SS, al igual que el PROFAD y el ESSDAI, como marcador de actividad. Se calculó el índice de confiabilidad del cuestionario en castellano con coeficiente de alfa de Cronbach y el coeficiente de correlación de Spearman para comparar las escalas. Se evaluaron también características demográficas, clínicas y de laboratorio.
ResultadosSe aplicó el ESSPRI, PROFAD y ESSDAI a 42 pacientes con SS, el 97.62% fueron mujeres. El resultado promedio del ESSPRI fue 5.8 (± 4.6), con un coeficiente de confiabilidad de 0.8034 y una correlación con el PROFAD de 0.5800 (p= 0.0001), y de -0.0848 (p=0,593) con el ESSDAI.
Discusión y conclusiones: La confiabilidad con la versión aplicada del ESSPRI en castellano fue adecuada. Se encontró una discordancia entre esta escala y el ESSDAI, lo cual remarca la importancia de aplicar ambas herramientas para asegurar un seguimiento objetivo del control de la enfermedad y su impacto en la calidad de vida de los pacientes con SS.
Sjögren’s syndrome (SS) is a systemic, chronic, autoimmune disease identified by its involvement of exocrine glands and presenting what is known as the “dry syndrome”, which may mainly be interpreted as the appearance of xerophthalmia and xerostomy. Up to 50% of patients with SS may present with other extra-glandular manifestations, such as musculoskeletal, pulmonary, gastrointestinal, hepatic, haematological, vascular, dermatological, renal and neurological symptoms.1 SS may be associated with other autoimmune diseases, such as rheumatoid arthritis to 20%–32%, systemic erythematosus lupus to 15%–36% and systemic sclerosis to 20%–32%, among others.2–4
SS affects the patients’ quality of life in physical, psychological and social spheres.5 For example, the symptoms such as dry eye, dry mouth, fatigue and joint pain are some of the many symptoms which have an impact on functionality and that may be disabilitating.6
Many clinimetric tools have been created over time, leading to the ability of the clinician to use scales to assess the activity, severity and damage of a disease and its impact on the patient’s quality of life.7 In the case of SS, questionnaires have been created, including, the Sicca Symptoms Inventory (SSI), to assess the dryness symptoms produced by SS8; the Profile of Fatigue and Discomfort (PROFAD), which focuses on the presence or absence of fatigue9; the Sjögren’s Systemic Clinical Activity Index’ (SCAI), which includes, among others, systemic manifestations.10 Recently, the EULAR Sjögren’s Syndrome Disease Activity Index (ESSDAI) and the EULAR Sjögren's Syndrome Patient Reported Index (ESSPRI)11 were validated. The ESSDAI is currently considered to be the gold standard for measuring disease activity,12 whilst the ESSPRI quantifies the severity of the debilitating symptoms of SS: pain, dryness and fatigue.13 The ESSPRI was designed in the year 2011, with appropriate correlation with scales, such as the PROFAD, the difference being that it is considerably simpler and less exhaustive than PROFAD.9,14
At present the ESSPRI is in English and has already been translated and validated into Portuguese.15 This study wished to carry out an adaptation of the ESSPRI from its original version into Spanish to facilitate its applicability in all SS patients in clinical studies and in clinical practice in Spanish-speaking countries.
MethodA cross-sectional study to validate the clinimetric scales in a centre of reference for autoimmune diseases, the Fundación Clínica Valle del Lili, en Cali, Colombia. Patients over 18 years of age were selected with classification of SS according to the 2016 American-European Consensus criteria,16 notwithstanding the coexistence or lack of it, of any other autoimmune disease (polyautoimmunity) or the treatment received. They had attended a check-up consultation for their disease at the Rheumatology Department between April 2018 and March 2019, in a single assessment. Exclusion criteria corresponded to those mentioned in the SS classification criteria (history of head and neck radiation, infection from active hepatitis virus, HIV infection, sarcoidosis, amyloidosis, graft-versus-host disease and igG4-associated disease).
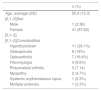
A cultural adaptation of the ESSPRI scale was made from the original version in English through translation-back translation, and the version of the scale were assessed by a review committee made up of specialist physicians with experience in disease management and/or scale management. The translation process was carried out with emphasis on the conceptual meaning of each item. After translation, the two versions were compared with regard to item significance.17,18 The approved version (Fig. 1) by the research group consisted of three statements relating to pain, fatigue and dryness in the previous two weeks, each with a possible score of between 0 and 10.
The ESSPRI and the PROFAD were applied to each patient as self-reporting, in the company of a researcher physician.
Demographic, clinical and laboratory data were collected from the medical file of the medical practice. Comorbidities which involved systemic compromise and/or the possibility of causing pain or fatigue were taken into consideration. The ESSDAI for each patient was calculated from the recorded information.
Sample size was defined from the criteria recommended in the literature:17,18 5 individuals per item of the tool to be assessed, i.e. 15 subjects as a minimum total for the study. However, our sample comprised 42 patients.
Statistical analysisA database with entry control was created through validation regulations of the scores introduced into the form, to guarantee reliability of data (Excel programme). After this, they were inserted into the Stata version 14.0 programme (StataCorp, College Station, Texas, U.S.A.), where data analysis was finally undertaken.
An exploratory data analysis was carried out, with normality of distribution, and extreme values which could affect the outcome, in addition to the lost values. After this a descriptive analysis of the study sample was performed, in keeping with the previously defined variables and means and medians were calculated according to the distribution found.
To establish internal consistency, Cronbach’s alpha coefficient was estimated for the scale items. To assess the construct validity the Spearman correlation coefficient was calculated for the ESSPRI with the PROFAD and for the ESSPRI with the ESSDAI.
ResultsForty two patients with a diagnosis of SS participated. The average age was 55.4years (±15.3), with a disease duration median of 36 (IQR 5.5-84) months ; 41 (97.62%) patients were women. Different comorbidities were found, with hypothyroidism being the most common (n=11. 26,19%), followed by osteoporosis (n=8, 19%) and osteoarthritis (n=7, 16.6%), among others; 18 (42.8%) patients presented with only one comorbidity, 6 presented with two (14.2%) and one with three (2.3%) (Table 1).
Demographic characteristics.
| n (%) | |
|---|---|
| Age, average (SD) | 55.4 (15.3) |
| [0,1–2]Sex | |
| Male | 1 (2.38) |
| Female | 41 (97.62) |
| [0,1–2] | |
| [0,1–2]Comorbidities | |
| Hypothyroidism | 11 (26.1%) |
| Osteoporosis | 8 (19%) |
| Osteoarthritis | 7 (16.6%) |
| Fibromyalgia | 4 (9.5%) |
| Rheumatoid arthritis | 3 (7.14) |
| Myopathy | 2 (4.7%) |
| Systemic erythematosus lupus | 1 (2.3%) |
| Multiple sclerosis | 1 (2.3%) |
The result of the ESSPRI for all patients was an average of 5.8 (±4.6) and for the PROFAD of 2.8 (±1.7). Furthermore, for the ESSDAI we found that 30 patients (71.43%) had low disease activity (median 0, IQR 0−0.75), 4 patients (9.52%) had moderate activity (median 8.5, IQR 6.5–9) and 8 patients (19.05%) had severe activity (average 33.1±15.30).
Standardised reliability coefficient (Cronbach’s alpha) of the ESSPRI was .8034 and Cronbach’s alpha of the PROFAD was .9623. Correlation between the ESSPRI and PROFAD scales was .5800 (p=.0001), whilst between the ESSPRI and the ESSDAI it was ―.0848 (p=.593) (Table 2).
Correlation between scales.
| Correlationa | p | |
|---|---|---|
| ESSPRI - PROFAD | .5800 | .0001 |
| ESSPRI - ESSDAI | ―.0848 | .593 |
The ESSPRI was validated in its original language in the year 2015 to assess the symptoms perceived by patients who had been diagnosed with SS.11,13 Since this index was based on three simple questions, it is a useful and user-friendly tool, and is therefore preferred over others like the PROFAD or the SSI, with which suitable correlation has been reported,13 and since then it has been used in several different clinical studies.19–21
The ESSPRI scale with its three domains translated into Spanish was used in this study in 42 patients diagnosed with SS. Compared with studies which only include patients with primary SS, our cohort contextualises on scenarios which may easily present in a rheumatology consultation, such as polyautoimmunity and even on infrequent associations, such as our case of multiple sclerosis and SS. Apart from this, several patients were found to present with one or more comorbidities. These included fibromyalgia, rheumatoid arthritis and hypothyroidism. All of these may generate pain and/or fatigue separately from the SS. Therefore, to differentiate the direct cause of these symptoms may be difficult and should be considered when this questionnaire is interpreted, without limiting its application. Linked to this is the fact that several studies have reported a correlation between disorders of anxiety and depression in patients with primary SS, not only with greater disease activity, but also with higher scores on the ESSPRI.19,22
Internal consistency of the test was high, based on the reliability coefficient for the ESSPRI of .8034, compared with .447 of adaptations into other languages.15 Correlation with the PROFAD was .58, similar to that obtained in the validation undertaken by the EULAR, of .68.11 In contrast to this, whilst in the initial validation of the scale a low correlation was found with the ESSDAI,11 in our case there was no correlation, given that r: ―.0848, with an insignificant p value.
Given that the ESSDAI is the tool of choice to discriminate between active and inactive disease by the clinician and the ESSPRI to assess the perception of symptoms by the patient, after demonstrating that the correlation between the two was low, it was suggested that the ESSPRI be used as complementary to the ESSDAI.11,23
Indeed, concordance between both scales is variable, depending on the context in which they are applied. Studies which measure the behaviour of the scale before and after administration of treatment in patients with SS have shown that the scores are concordant with reduction and lastly that there is sufficient sensitivity in these scales for change to occur after an intervention.24,25 In the Olsson et al.26 series, the ESSPRI was associated with higher accumulated consumption of tobacco, whilst the ESSDAI did not show there to be any relationship; even so, when measuring them according to the state of the patients’ tobacco habit, the two behaved similarly. In our cohort we were able to see that several patients had low scores in the ESSDAI and high scores in the ESSPRI, which shows a discrepancy between the point of view of the clinician and the patient on the disease at a specific moment in time, comparable with other reports in which discordance between the two scales was found.21
Furthermore, although the application of the ESSPRI is aimed at subjective assessment of symptoms, several studies have described a correlation with biological variables such as serum levels of beta-2 microglobulin and erythrocyte sedimentation rate.20,27
Despite the fact that the ESSPRI has been incorporated as a marker of outcomes and suchlike in research studies, little is to be found in the literature on its use by rheumatologists in their daily clinical practice.
The aim of SS management is steady control of the disease with minimum activity and to guarantee the patient quality of life. Ideally, therefore, it should be applied jointly with the ESSDAI for a better reflection of the entire spectrum of the disease and even more so because they are currently the clinometric scales classified as the most practical and reliable in SS.28
Although this adaptation into Spanish was conducted with Colombian patients and its application into other Spanish-speaking countries requires studied evaluation, it is expected to be of great use for implementation, follow-up and decision-making in these countries.
Study limitationsNo intraobserver reproducibility test was conducted, in contrast to other studies which assess clinimetric scales. No strict evaluation was made of the possible difficulties the patient may have had when completing it and/or the tests, which should be taken into consideration in its implementation.
FinancingNo financial support was received for this study.
Conflict of interestsThe authors have no conflict of interests to declare.
Please cite this article as: Posso-Osorio I, Nieto-Aristizábal I, Soto D, Ariza C, Urbano M, Cañas CA, et al. Validación y adaptación al castellano del Índice Reportado por Pacientes con Síndrome de Sjögren del EULAR (ESSPRI-EULAR Sjögren’s Syndrome Patient Reported Index). Reumatol Clin. 2021;17:388–391.