Sarcoidosis is a chronic, inflammatory disease with unknown cause characterized by non-caseating granuloma formations. It can be presented with bilateral hilar lymphadenopathy, skin lesions, eye involvement and locomotor system findings. Adult onset Still disease (AOSD) is a chronic inflammatory disease which presents with fever, arthritis and typical skin rashes. The disease is rare and can be misdiagnosed due to the absence of typical clinical and laboratory findings. The association of sarcoidosis and AOSD has not been previously reported in the literature.
Herein we reported the development of AOSD in a patient followed by the diagnosis of sarcoidosis. The patient did not respond to high-dose corticosteroids and methotrexate therapy, and the disease was under control with anti-IL-6 (Tocilizumab) drug.
La sarcoidosis es una enfermedad inflamatoria crónica de causa desconocida, que se caracteriza por formaciones de granulomas no caseificantes. Puede presentarse como linfadenopatía hiliar bilateral, lesiones cutáneas, compromiso ocular y del sistema locomotor. La enfermedad de Still del adulto (AOSD) es una enfermedad inflamatoria crónica que se presenta con fiebre, artritis y erupciones cutáneas típicas. Dicha enfermedad es rara y puede diagnoticarse erróneamente debido a la ausencia de rasgos clínicos y de laboratorio típicos. En la literatura no se ha reportado previamente la asociación entre sarcoidosis y AOSD.
Reportamos aquí el desarrollo de AOSD en un paciente sometido a seguimiento debido a diagnóstico de sarcoidosis. El paciente no respondió al tratamiento con altas dosis de corticosteroides y metotrexato, manteniéndose la enfermedad bajo control con un fármaco anti-IL-6 (Tocilizumab).
Sarcoidosis is a systemic disease of unknown origin characterized by a non-caseating granulomatous reaction that can involves many tissues and organs.1 Although the pathogenesis is not clear yet, some genetic and environmental factors influence the cellular immune system activation and non-specific inflammatory response occurred. T-helper 1 and proinflammatory cytokines from macrophages trigger inflammatory cascade and granuloma formation may occur as a result of tissue permeability, cell influx, and local cell proliferation. Presence of non-caseating epithelioid cell granulomas is the indisputable pathological finding of sarcoidosis.2 Different prevalence, clinical signs and disease course in different ethnic and racial groups are indicative for sarcoidosis as being a heterogeneous disease.3 Its prevalence is high in female and it generally occurs at age of 40. The incidence of sarcoidosis in USA is 10.9/100.000 in white, while in Afro-American this rate is 35.5/100.000.4
Adult onset Still's disease (AOSD) is a rare systemic inflammatory disease characterized by high fever, transient skin rash, arthritis and many organ involvement.5 The incidence of the disease is 1–10/1mln, the ratio of female to male is 2/1, and it is seen most frequently in 16–35 years of age. Pathogenesis is not elucidated yet, but it is thought to be a Th1-related disease. As intensive cytokine release, fever, rash and association of serositis are similar characteristics of AOSD with autoinflammatory diseases; there are those who advocate taking part of AOSD in this group of diseases.6 There are insufficient data in the literature about the association of sarcoidosis and AOSD.
Herein we reported the coexistence of sarcoidosis and AOSD.
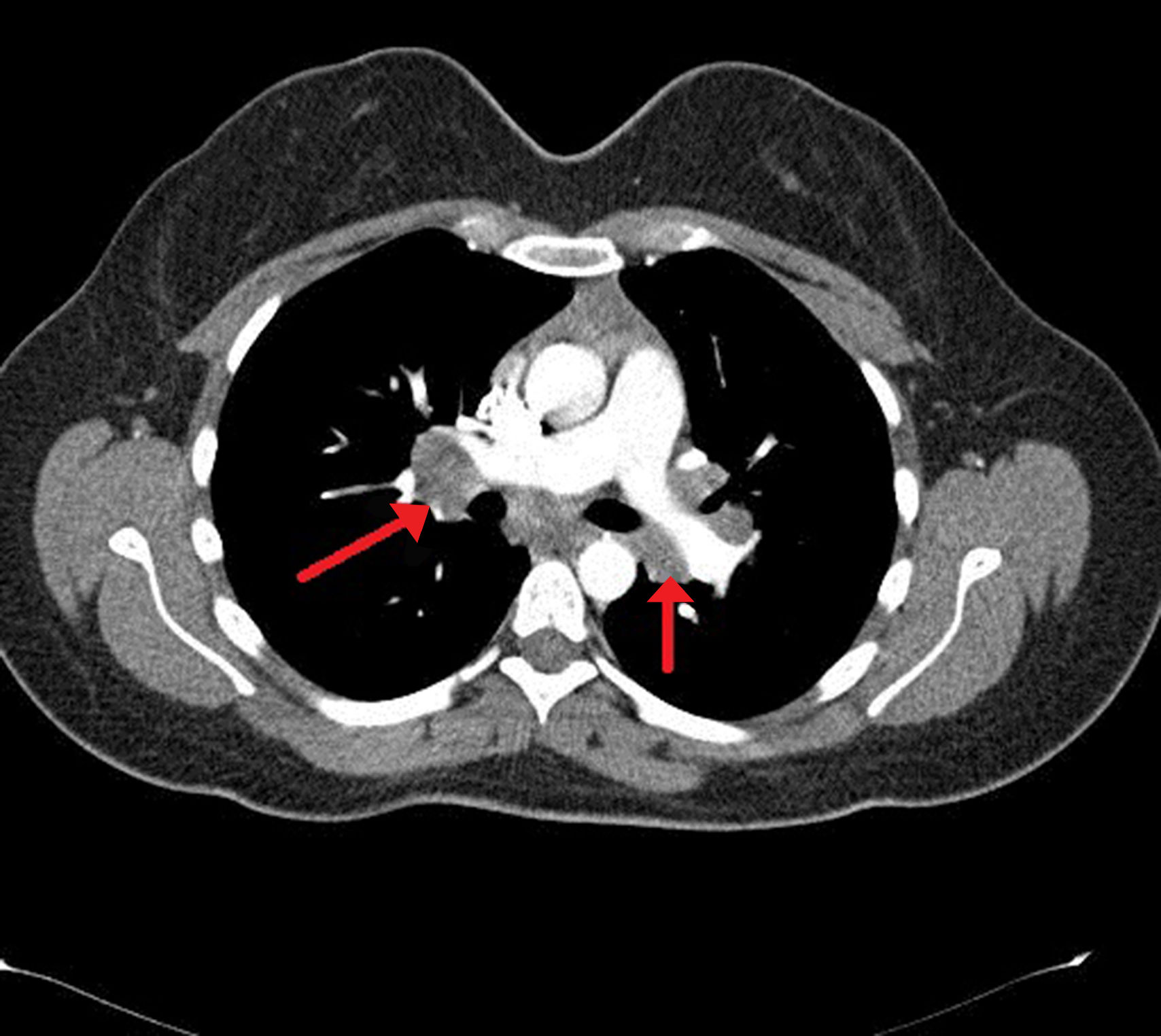
Case presentationA 24-year-old male patient admitted to the Rheumatology polyclinic with complaints of arthritis of both ankle joints, erythema nodosum and fatigue for about 4 years ago. Chest disease consultation was made due to acute phase reactant elevation and detection of bilateral hilar lymphadenopathy at thorax computed tomography (CT) (Fig. 1). Non-caseating granuloma was detected in endobronchial ultrasound (EBUS) biopsy which was made with sarcoidosis pre-diagnosis. Sarcoidosis was diagnosed also due to high CD4/CD8 ratio in the bronchoalveolar lavage (BAL) fluid, typical thorax CT images and biopsy results. The disease was controlled by low-dose corticosteroid therapy and drug was discontinued after about 1 year. Six months after the treatment was discontinued, patient applied to the Rheumatology polyclinic because of complaints of fever, diffuse maculopapular skin rashes, joint pain and morning stiffness. In physical examination; fever 39°C, bilateral axillary lymphadenopathy, mild hepatosplenomegaly, widespread skin rash, tenderness and limitation of motion in knee, wrist, metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints were detected. Laboratory tests revealed acute phase reactants and ferritin elevation. Serological examinations including rheumatoid factor (RF), anti-nuclear antibody (ANA), anti-cytoplasmic antibody (ANCA), anti-cyclic citrullinated antibody (anti-CCP) were normal. Due to the fever, an infectious disease specialist consultation was made for possible infection, no infection focus was detected. Blood and urine culture results were negative. Viral serologic tests (EBV, CMV, HIV, HBV, HCV) and Brucella tests were normal. Peripheral blood smear was performed, everything was normal except neutrophilic leukocytosis. Hematology consultation was performed, hematological malignancy was not considered. Mild hepatosplenomegaly was detected in abdominal ultrasonography (USG) and axillary USG showed lymphadenopathy with 5–6 lymph nodes and the largest one was 12×4mm in diameter. Chest X-ray and thorax CT were normal. There were no findings suggestive of sarcoidosis activation. According to clinical, laboratory and serological tests the patient was diagnosed with AOSD and corticosteroids (CS)1mg/kg/day plus methotrexate (MTX) 15mg/week were started. After 3 months, there was a slight decrease in acute phase reactants, but there was no significant decline in patient complaints and MTX dose was increased to 20mg/week. After 5months of treatment Tocilizumab was started in a rheumatology center to the patient who applied without any significant regression in his complaints. Significant regression was observed in the patient's complaints at 6th month of this treatment, acute phase reactants and serum ferritin levels were normalized. Policlinic follow-up of the patient with good general condition and remission of the disease is continuing.
DiscussionIn this paper, we reported the coexistence of sarcoidosis and AOSD. Our patient, who was followed with diagnosis of sarcoidosis, presenting with complaints of fever, skin rash and arthritis and initially was hospitalized for sarcoidosis reactivation. However, because of the presence of rapidly fading maculopapular eruptions associated with fever, lymphadenopathy, hepatosplenomegaly, elevated liver function tests and ferritin, and after the exclusion of another underlying pathology. Our patient was diagnosed as AOSD. Patient was not responding to high doses CS and MTX, disease was controlled after Tocilizumab treatment.
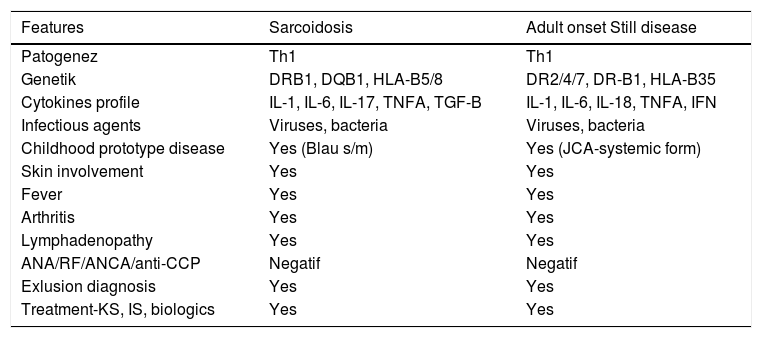
Some similar features of sarcoidosis and AOSD are available (Table 1). Etiopathogenesis of both diseases is unclear, but they result in a Th1-weighted immune response at the genetic basis, with the effect of some environmental factors and infectious agents. The presence of prototypes (Blau s/m and juvenile chronic arthritis systemic form) in childhood, and similar cytokine profiles and similar clinical features (such as fever, skin and joint involvement, lymphadenopathy and multiorgan involvement) are some of the common features of both diseases. In addition, it is similar between both diseases that diagnosis of both disease can be made just after the exclusion of other diseases (such as infection, malignancy, other rheumatic diseases).
Sarcoidosis vs. adult onset Still's disease – common similarities.
| Features | Sarcoidosis | Adult onset Still disease |
|---|---|---|
| Patogenez | Th1 | Th1 |
| Genetik | DRB1, DQB1, HLA-B5/8 | DR2/4/7, DR-B1, HLA-B35 |
| Cytokines profile | IL-1, IL-6, IL-17, TNFA, TGF-B | IL-1, IL-6, IL-18, TNFA, IFN |
| Infectious agents | Viruses, bacteria | Viruses, bacteria |
| Childhood prototype disease | Yes (Blau s/m) | Yes (JCA-systemic form) |
| Skin involvement | Yes | Yes |
| Fever | Yes | Yes |
| Arthritis | Yes | Yes |
| Lymphadenopathy | Yes | Yes |
| ANA/RF/ANCA/anti-CCP | Negatif | Negatif |
| Exlusion diagnosis | Yes | Yes |
| Treatment-KS, IS, biologics | Yes | Yes |
Abbreviations: IL – interleukine; TNFA – tumor necrosis factor alpha; TGF-B – transforming growth factor beta; JCA – juvenile chronic arthritis, IFN – interferon; HLA – human leukocyte antigen; Th1 – T-helper 1; ANA – antinuclear antibody; RF – rheumatoid factor.
AOSD is a rare systemic inflammatory disease characterized by high fever, transient maculopapular skin rash, arthritis and many organ involvement.7 Although the pathogenesis of the disease is unclear, it is thought that it develops as a result of the immune system dysfunction in which Th1 cells plays important role.8 Some infectious agents are also accused but no specific agent has been identified in cultures. HLA DR2, DR4, DR7 and HLA B35 positivity is frequently seen in this patients.9 AOSD is an exclusionary diagnosis, so the differential diagnosis must be done very well. First of all, infectious diseases, malignancies and other rheumatic diseases that make this clinical picture should be excluded. In distinguishing different connective tissue diseases, ANA and RF negativity are important. Besides significantly elevated ferritin levels are also helpful in diagnosis. For this reason, it is recommended that the patient should be examined for serum ferritin level, especially if there are any rheumatologic findings.10 As in our patients, high levels of ferritin can be a guide. Since the AOSD does not have a specific laboratory finding and is not considered clinically preliminary, the diagnosis is either skipped or diagnosed late. Diagnostic criteria have been developed by different investigators and tested for their sensitivity and specificity. The diagnostic criteria developed by Yamaguchi were reported to be 96% sensitive and 92% specific. According to this criteria diagnosis can be made with more than 5 criteria, 2 of which are major.11 In our patient, three of the major criteria and four of the minor criteria were present. In the light of this data, our patient was diagnosed and treated as AOSD. The disease should be treated according to the severity of the disease and organ involvement. Although nonsteroidal anti-inflammatory drugs (NSAIDs) are recommended in the first place, they have a very low effect and CSs and immunosuppressive drugs (MTX, azathioprin, cyclosporin A) have been shown to be more effective.12–14 CS doses during an acute exacerbation is usually 1mg/kg/day and it is effective in many patients. Pulsed methyl prednisolone is used in severe hepatic involvement, cardiac tamponade, disseminated intravascular coagulation (DIC) or other life-threatening conditions.15 The effects of biological agents have been shown in patients unresponsive to CS and immunosuppressive treatment.16–18 Since our case was not responded to high dose CS and MTX treatment, Tocilizumab was started. With this treatment, the disease is under control and it is now in remission.
In conclusion, we reported the development of AOSD in a patient with a diagnosis of sarcoidosis. Since sarcoidosis and AOSD have some similar characteristics, differential diagnosis must be performed well. When symptoms such as fever, arthritis, skin rash developed in patients with sarcoidosis, AOSD should be considered in addition to activation of sarcoidosis. The coexistence of these two diseases, which are imitators of many different diseases, can be a coincidence and/or they might have a common etiopathogenesis. More research is needed in this regard.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestsThe authors declare no conflict of interest.